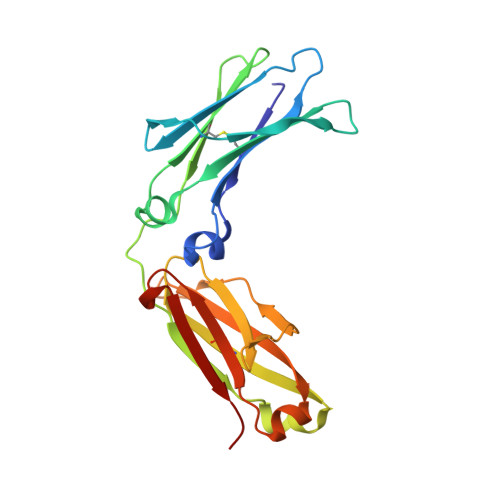
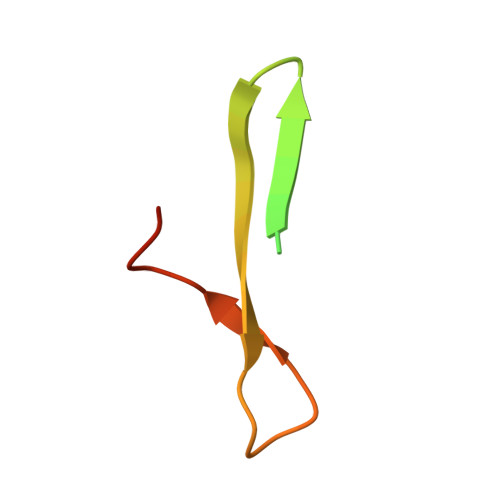
Adaptive Assembly: Maximizing the Potential of a Given Functional Peptide with a Tailor-Made Protein Scaffold.
Watanabe, H., Honda, S.(2015) Chem Biol 22: 1165-1173
- PubMed: 26299673
- DOI: https://doi.org/10.1016/j.chembiol.2015.07.015
- Primary Citation of Related Structures:
3WKN - PubMed Abstract:
Protein engineering that exploits known functional peptides holds great promise for generating novel functional proteins. Here we propose a combinatorial approach, termed adaptive assembly, which provides a tailor-made protein scaffold for a given functional peptide. A combinatorial library was designed to create a tailor-made scaffold, which was generated from β hairpins derived from a 10-residue minimal protein "chignolin" and randomized amino acid sequences. We applied adaptive assembly to a peptide with low affinity for the Fc region of human immunoglobulin G, generating a 54-residue protein AF.p17 with a 40,600-fold enhanced affinity. The crystal structure of AF.p17 complexed with the Fc region revealed that the scaffold fixed the active conformation with a unique structure composed of a short α helix, β hairpins, and a loop-like structure. Adaptive assembly can take full advantage of known peptides as assets for generating novel functional proteins.
Organizational Affiliation:
Biomedical Research Institute, the National Institute of Advanced Industrial Science and Technology (AIST), 1-1-1, Higashi, Tsukuba, Ibaraki 305-8566, Japan.