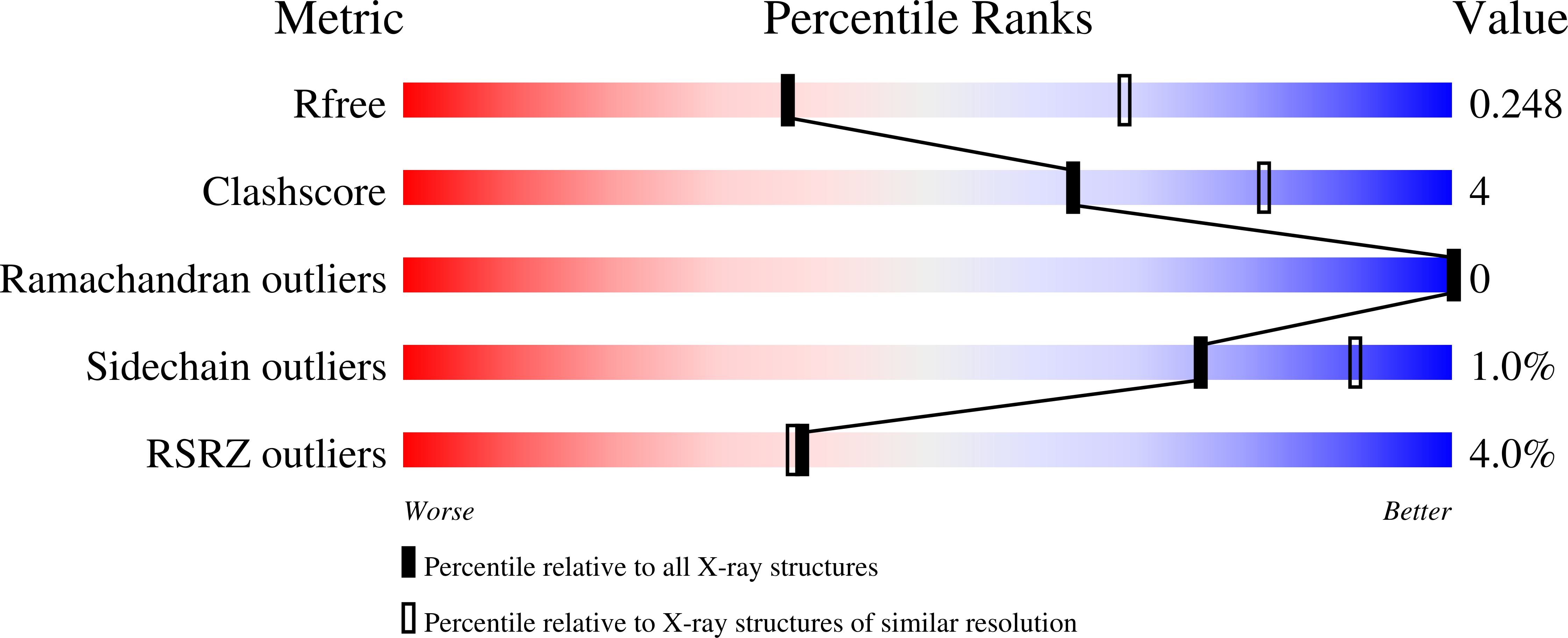
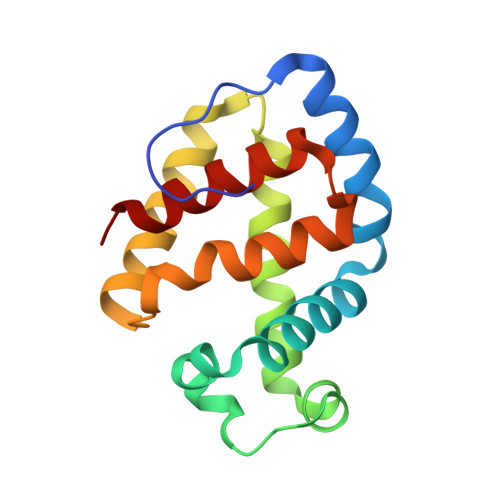
The 2.7 A crystal structure of deoxygenated hemoglobin from the sea lamprey (Petromyzon marinus): structural basis for a lowered oxygen affinity and Bohr effect.
Heaslet, H.A., Royer Jr., W.E.(1999) Structure 7: 517-526
- PubMed: 10378271
- DOI: https://doi.org/10.1016/s0969-2126(99)80068-9
- Primary Citation of Related Structures:
3LHB - PubMed Abstract:
The hemoglobins of the sea lamprey are unusual in that cooperativity and sensitivity to pH arise from an equilibrium between a high-affinity monomer and a low-affinity oligomer. Although the crystal structure of the monomeric cyanide derivative has previously been determined, the manner by which oligomerization acts to lower the oxygen affinity and confer a strong Bohr effect has, until now, been speculative.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, University of Massachusetts Medical School, Worcester 01655, USA.