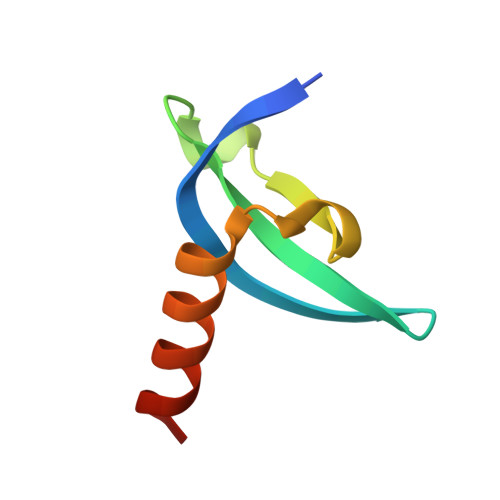
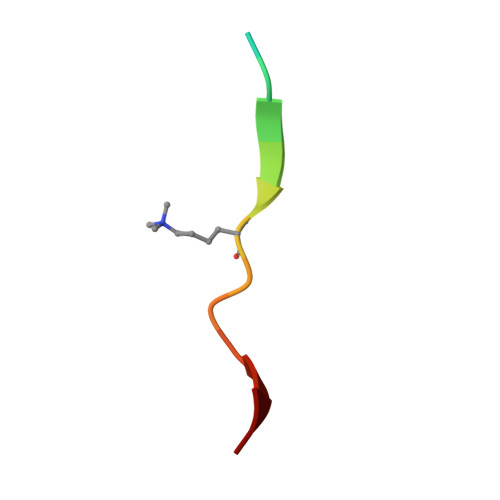
Structural basis for specific binding of human MPP8 chromodomain to histone H3 methylated at lysine 9.
Li, J., Li, Z., Ruan, J., Xu, C., Tong, Y., Pan, P.W., Tempel, W., Crombet, L., Min, J., Zang, J.(2011) PLoS One 6: e25104-e25104
- PubMed: 22022377
- DOI: https://doi.org/10.1371/journal.pone.0025104
- Primary Citation of Related Structures:
3LWE, 3R93 - PubMed Abstract:
M-phase phosphoprotein 8 (MPP8) was initially identified to be a component of the RanBPM-containing large protein complex, and has recently been shown to bind to methylated H3K9 both in vivo and in vitro. MPP8 binding to methylated H3K9 is suggested to recruit the H3K9 methyltransferases GLP and ESET, and DNA methyltransferase 3A to the promoter of the E-cadherin gene, mediating the E-cadherin gene silencing and promote tumor cell motility and invasion. MPP8 contains a chromodomain in its N-terminus, which is used to bind the methylated H3K9.
Organizational Affiliation:
Key Laboratory of Structural Biology, Chinese Academy of Sciences, and School of Life Sciences, University of Science and Technology of China, Hefei, Anhui, People's Republic of China.