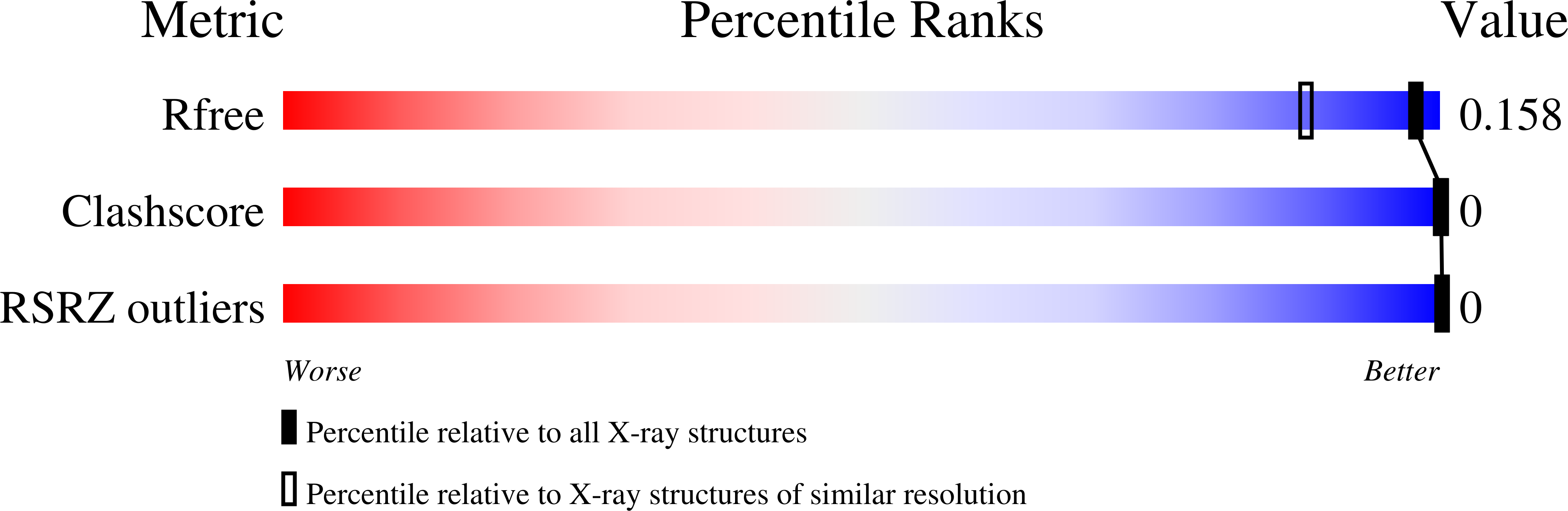An exocyclic methylene group acts as a bioisostere of the 2'-oxygen atom in LNA.
Seth, P.P., Allerson, C.R., Berdeja, A., Siwkowski, A., Pallan, P.S., Gaus, H., Prakash, T.P., Watt, A.T., Egli, M., Swayze, E.E.(2010) J Am Chem Soc 132: 14942-14950
- PubMed: 20886816
- DOI: https://doi.org/10.1021/ja105875e
- Primary Citation of Related Structures:
3OZ3, 3OZ4, 3OZ5 - PubMed Abstract:
We show for the first time that it is possible to obtain LNA-like (Locked Nucleic Acid 1) binding affinity and biological activity with carbocyclic LNA (cLNA) analogs by replacing the 2'-oxygen atom in LNA with an exocyclic methylene group. Synthesis of the methylene-cLNA nucleoside was accomplished by an intramolecular cyclization reaction between a radical at the 2'-position and a propynyl group at the C-4' position. Only methylene-cLNA modified oligonucleotides showed similar thermal stability and mismatch discrimination properties for complementary nucleic acids as LNA. In contrast, the close structurally related methyl-cLNA analogs showed diminished hybridization properties. Analysis of crystal structures of cLNA modified self-complementary DNA decamer duplexes revealed that the methylene group participates in a tight interaction with a 2'-deoxyribose residue of the 5'-terminal G of a neighboring duplex, resulting in the formation of a CH...O type hydrogen bond. This indicates that the methylene group retains a negative polarization at the edge of the minor groove in the absence of a hydrophilic 2'-substituent and provides a rationale for the superior thermal stability of this modification. In animal experiments, methylene-cLNA antisense oligonucleotides (ASOs) showed similar in vivo activity but reduced toxicity as compared to LNA ASOs. Our work highlights the interchangeable role of oxygen and unsaturated moieties in nucleic acid structure and emphasizes greater use of this bioisostere to improve the properties of nucleic acids for therapeutic and diagnostic applications.
Organizational Affiliation:
Department of Medicinal Chemistry, Isis Pharmaceuticals, Inc., 1891 Rutherford Road, Carlsbad, California 92008, USA. pseth@isisph.com