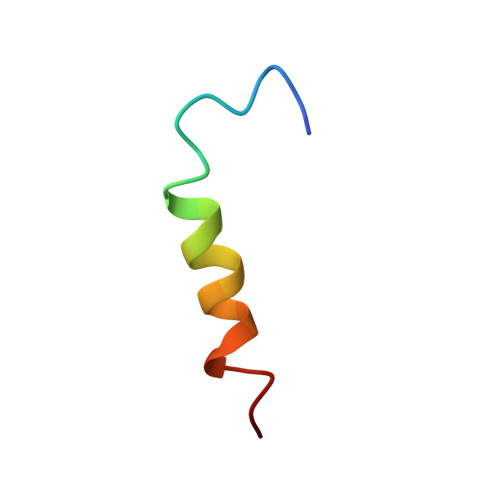
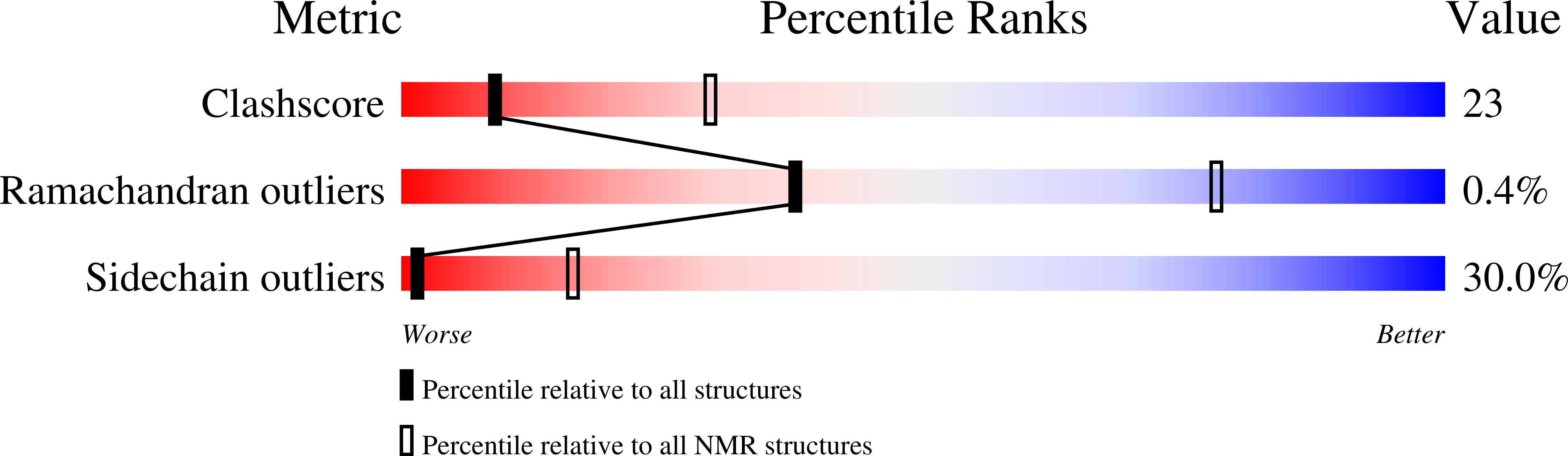Structural properties of a peptide derived from H(+)-V-ATPase subunit a
Vermeer, L.S., Reat, V., Hemminga, M.A., Milon, A.(2009) Biochim Biophys Acta 1788: 1204-1212
- PubMed: 19249284
- DOI: https://doi.org/10.1016/j.bbamem.2009.02.015
- Primary Citation of Related Structures:
2RPW - PubMed Abstract:
The 3D structure of a peptide derived from the putative transmembrane segment 7 (TM7) of subunit a from H(+)-V-ATPase from Saccharomyces cerevisiae has been determined by solution state NMR in SDS. A stable helix is formed from L736 up to and including Q745, the lumenal half of the putative TM7. The helical region extends well beyond A738, as was previously suggested based on NMR studies of a similar peptide in DMSO. The pKa of both histidine residues that are important for proton transport was measured in water and in SDS. The differences that are found demonstrate that the histidine residues interact with the SDS polar heads. In detergent, circular dichroism data indicate that the secondary structure of the peptide depends on the pH and the type of detergent used. Using solid-state NMR, it is shown that the peptide is immobile in phospholipid bilayers, which means that it is probably not a single transmembrane helix in these samples. The environment is important for the structure of TM7, so in subunit a it is probably held in place by the other transmembrane helices of this subunit.
Organizational Affiliation:
Université de Toulouse, UPS, IPBS, 205 route de Narbonne, 31077 Toulouse Cedex, France.