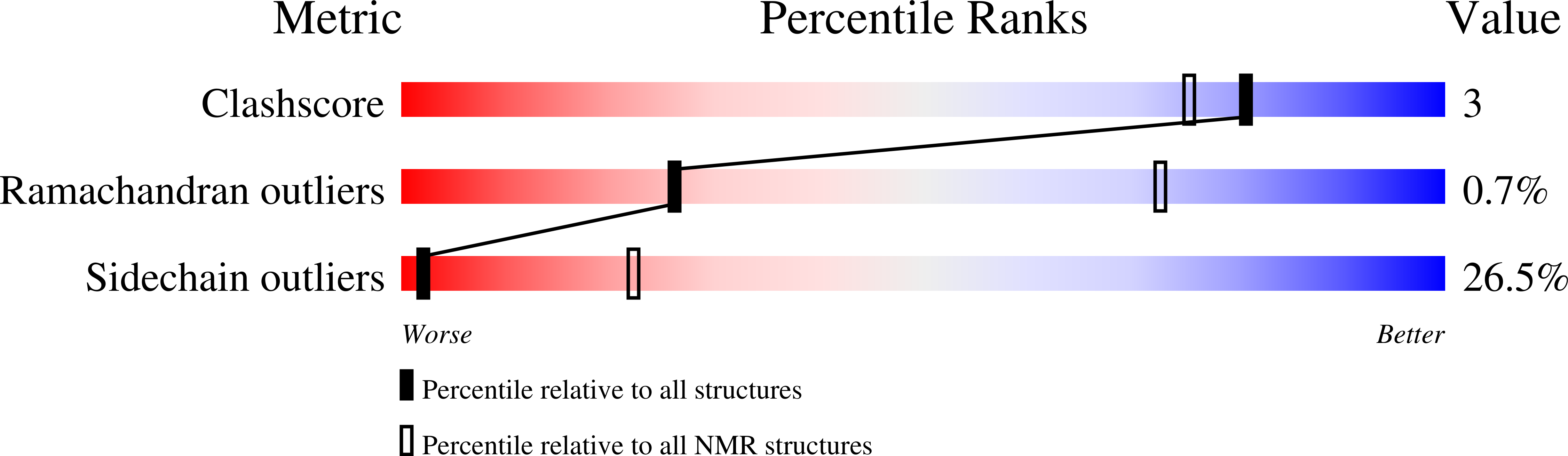Structural Basis for a Novel Interaction between the NS1 Protein Derived from the 1918 Influenza Virus and RIG-I.
Jureka, A.S., Kleinpeter, A.B., Cornilescu, G., Cornilescu, C.C., Petit, C.M.(2015) Structure 23: 2001-2010
- PubMed: 26365801
- DOI: https://doi.org/10.1016/j.str.2015.08.007
- Primary Citation of Related Structures:
2N74 - PubMed Abstract:
The influenza non-structural protein 1 (NS1) plays a critical role in antagonizing the innate immune response to infection. One interaction that facilitates this function is between NS1 and RIG-I, one of the main sensors of influenza virus infection. While NS1 and RIG-I are known to interact, it is currently unclear whether this interaction is direct or if it is mediated by other biomolecules. Here we demonstrate a direct, strain-dependent interaction between the NS1 RNA binding domain (NS1(RBD)) of the influenza A/Brevig Mission/1918 H1N1 (1918(H1N1)) virus and the second caspase activation and recruitment domain of RIG-I. Solving the solution structure of the 1918(H1N1) NS1(RBD) revealed features in a functionally novel region that may facilitate the observed interaction. The biophysical and structural data herein suggest a possible mechanism by which strain-specific differences in NS1 modulate influenza virulence.
Organizational Affiliation:
Department of Biochemistry and Molecular Genetics, University of Alabama at Birmingham, Birmingham, AL 35294, USA.