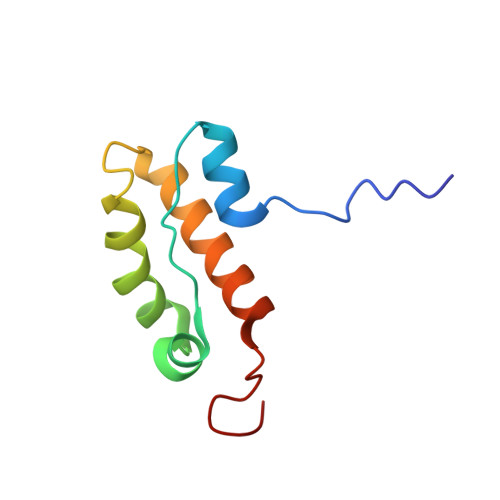
The N-terminal Region of Chromodomain Helicase DNA-binding Protein 4 (CHD4) Is Essential for Activity and Contains a High Mobility Group (HMG) Box-like-domain That Can Bind Poly(ADP-ribose).
Silva, A.P., Ryan, D.P., Galanty, Y., Low, J.K., Vandevenne, M., Jackson, S.P., Mackay, J.P.(2016) J Biol Chem 291: 924-938
- PubMed: 26565020
- DOI: https://doi.org/10.1074/jbc.M115.683227
- Primary Citation of Related Structures:
2N5N - PubMed Abstract:
Chromodomain Helicase DNA-binding protein 4 (CHD4) is a chromatin-remodeling enzyme that has been reported to regulate DNA-damage responses through its N-terminal region in a poly(ADP-ribose) polymerase-dependent manner. We have identified and determined the structure of a stable domain (CHD4-N) in this N-terminal region. The-fold consists of a four-α-helix bundle with structural similarity to the high mobility group box, a domain that is well known as a DNA binding module. We show that the CHD4-N domain binds with higher affinity to poly(ADP-ribose) than to DNA. We also show that the N-terminal region of CHD4, although not CHD4-N alone, is essential for full nucleosome remodeling activity and is important for localizing CHD4 to sites of DNA damage. Overall, these data build on our understanding of how CHD4-NuRD acts to regulate gene expression and participates in the DNA-damage response.
Organizational Affiliation:
From the School of Molecular Bioscience, The University of Sydney, New South Wales 2006, Australia, ana.silva@sydney.edu.au.