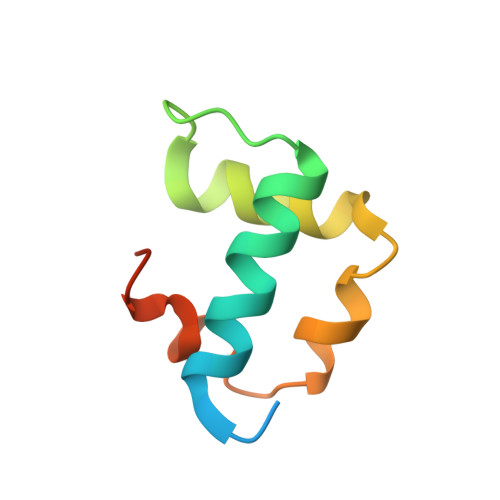
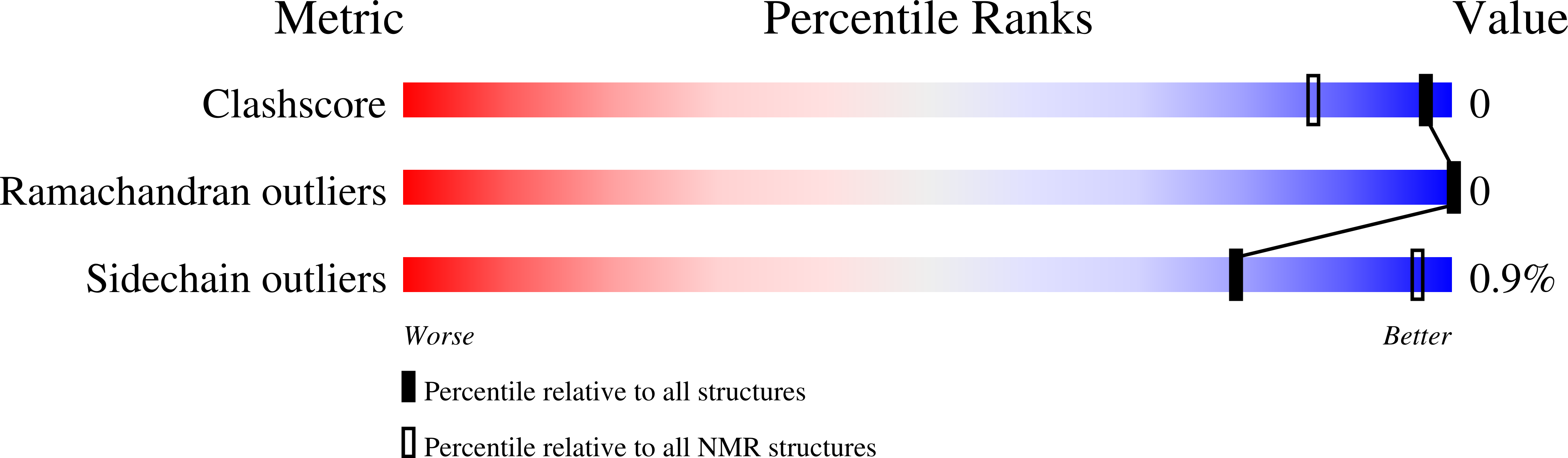A transient and low-populated protein-folding intermediate at atomic resolution.
Korzhnev, D.M., Religa, T.L., Banachewicz, W., Fersht, A.R., Kay, L.E.(2010) Science 329: 1312-1316
- PubMed: 20829478
- DOI: https://doi.org/10.1126/science.1191723
- Primary Citation of Related Structures:
2KZG - PubMed Abstract:
Proteins can sample conformational states that are critical for function but are seldom detected directly because of their low occupancies and short lifetimes. In this work, we used chemical shifts and bond-vector orientation constraints obtained from nuclear magnetic resonance relaxation dispersion spectroscopy, in concert with a chemical shift-based method for structure elucidation, to determine an atomic-resolution structure of an "invisible" folding intermediate of a small protein module: the FF domain. The structure reveals non-native elements preventing formation of the native conformation in the carboxyl-terminal part of the protein. This is consistent with the kinetics of folding in which a well-structured intermediate forms rapidly and then rearranges slowly to the native state. The approach introduces a general strategy for structure determination of low-populated and transiently formed protein states.
Organizational Affiliation:
Department of Molecular Genetics, the University of Toronto, Toronto, Ontario M5S 1A8, Canada.