Protein Crystallography with a Micrometre-Sized Synchrotron-Radiation Beam.
Moukhametzianov, R., Burghammer, M., Edwards, P.C., Petitdemange, S., Popov, D., Fransen, M., Mcmullan, G., Schertler, G.F., Riekel, C.(2008) Acta Crystallogr D Biol Crystallogr 64: 158
- PubMed: 18219115
- DOI: https://doi.org/10.1107/S090744490705812X
- Primary Citation of Related Structures:
2JIC - PubMed Abstract:
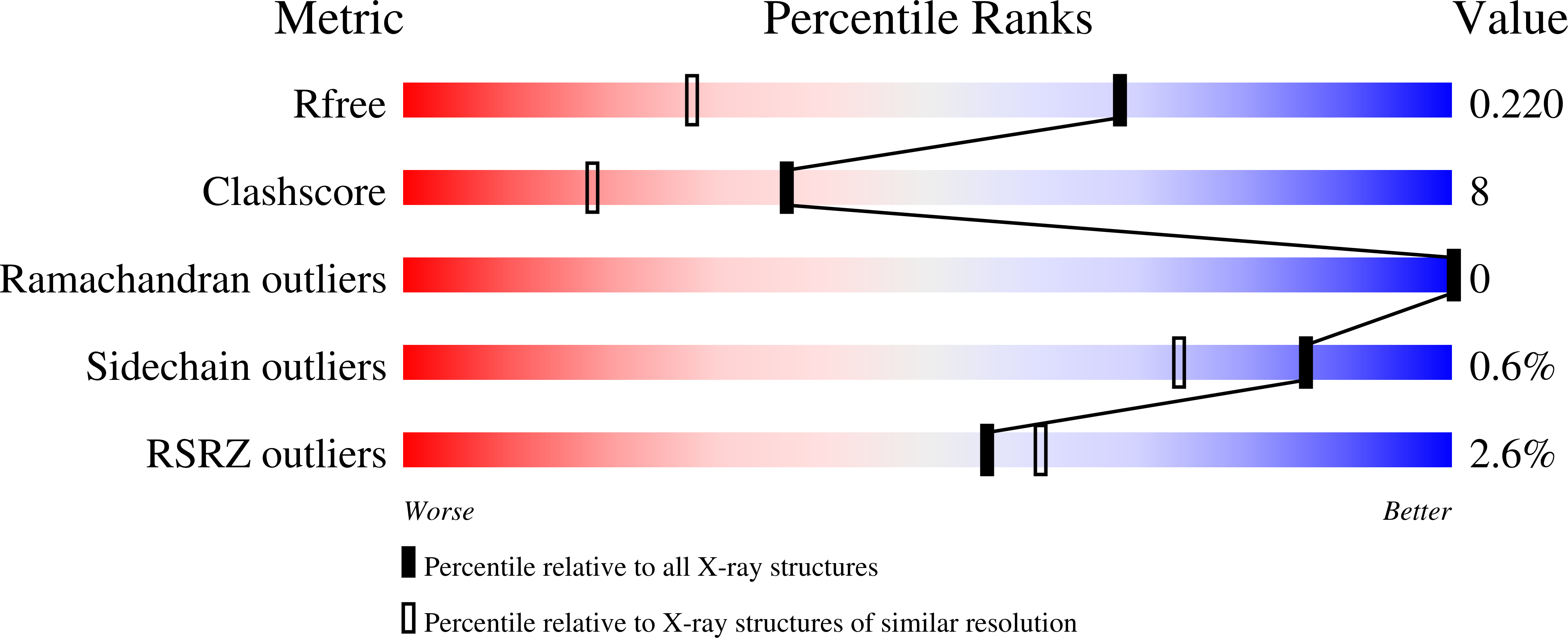
For the first time, protein microcrystallography has been performed with a focused synchrotron-radiation beam of 1 microm using a goniometer with a sub-micrometre sphere of confusion. The crystal structure of xylanase II has been determined with a flux density of about 3 x 10(10) photons s(-1) microm(-2) at the sample. Two sets of diffraction images collected from different sized crystals were shown to comprise data of good quality, which allowed a 1.5 A resolution xylanase II structure to be obtained. The main conclusion of this experiment is that a high-resolution diffraction pattern can be obtained from 20 microm(3) crystal volume, corresponding to about 2 x 10(8) unit cells. Despite the high irradiation dose in this case, it was possible to obtain an excellent high-resolution map and it could be concluded from the individual atomic B-factor patterns that there was no evidence of significant radiation damage. The photoelectron escape from a narrow diffraction channel is a possible reason for reduced radiation damage as indicated by Monte Carlo simulations. These results open many new opportunities in scanning protein microcrystallography and make random data collection from microcrystals a real possibility, therefore enabling structures to be solved from much smaller crystals than previously anticipated as long as the crystallites are well ordered.
Organizational Affiliation:
MRC Laboratory of Molecular Biology, Hills Road, Cambridge CB2 0QH, England.