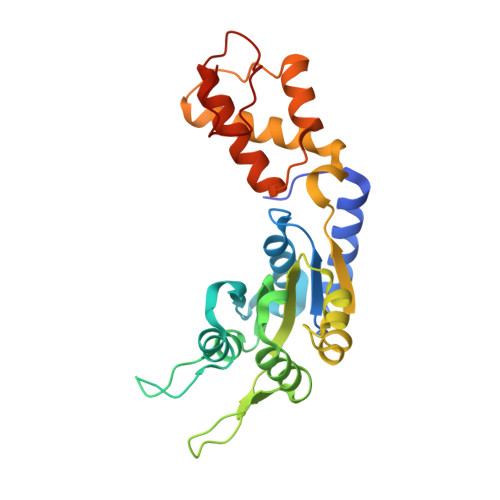
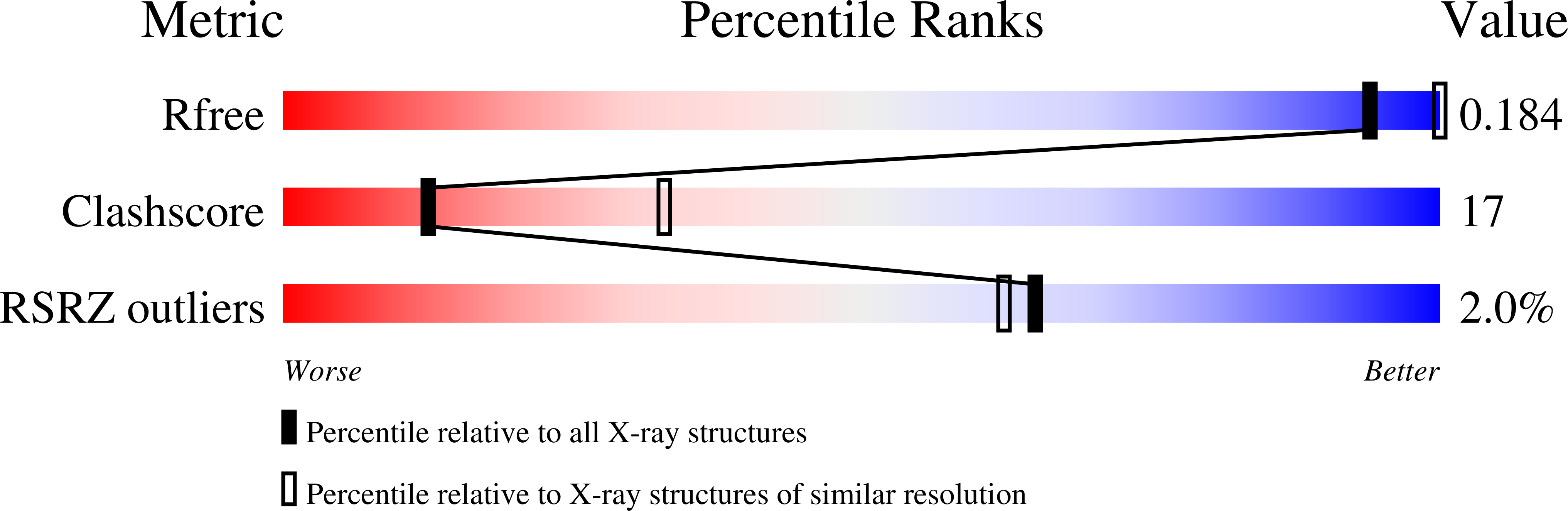Trapping of a Transcription Complex Using a New Nucleotide Analogue: AMP Aluminium Fluoride
Joly, N., Rappas, M., Buck, M., Zhang, X.(2008) J Mol Biol 375: 1206
- PubMed: 18082766
- DOI: https://doi.org/10.1016/j.jmb.2007.11.050
- Primary Citation of Related Structures:
2VII - PubMed Abstract:
Mechanochemical proteins rely on ATP hydrolysis to establish the different functional states required for their biological output. Studying the transient functional intermediate states these proteins adopt as they progress through the ATP hydrolysis cycle is key to understanding the molecular basis of their mechanism. Many of these intermediates have been successfully 'trapped' and functionally characterised using ATP analogues. Here, we present a new nucleotide analogue, AMP-AlF(x), which traps PspF, a bacterial enhancer binding protein, in a stable complex with the sigma(54)-RNA polymerase holoenzyme. The crystal structure of AMP-AlF(x)*PspF(1-275) provides new information on protein-nucleotide interactions and suggests that the beta and gamma phosphates are more important than the alpha phosphate in terms of sensing nucleotide bound states. In addition, functional data obtained with AMP-AlF(x) establish distinct roles for the conserved catalytic AAA(+) (ATPases associated with various cellular activities) residues, suggesting that AMP-AlF(x) is a powerful new tool to study AAA(+) protein family members and, more generally, Walker motif ATPases.
Organizational Affiliation:
Division of Biology, Sir Alexander Fleming Building, Imperial College London, Exhibition Road, South Kensington, London SW7 2AZ, UK.