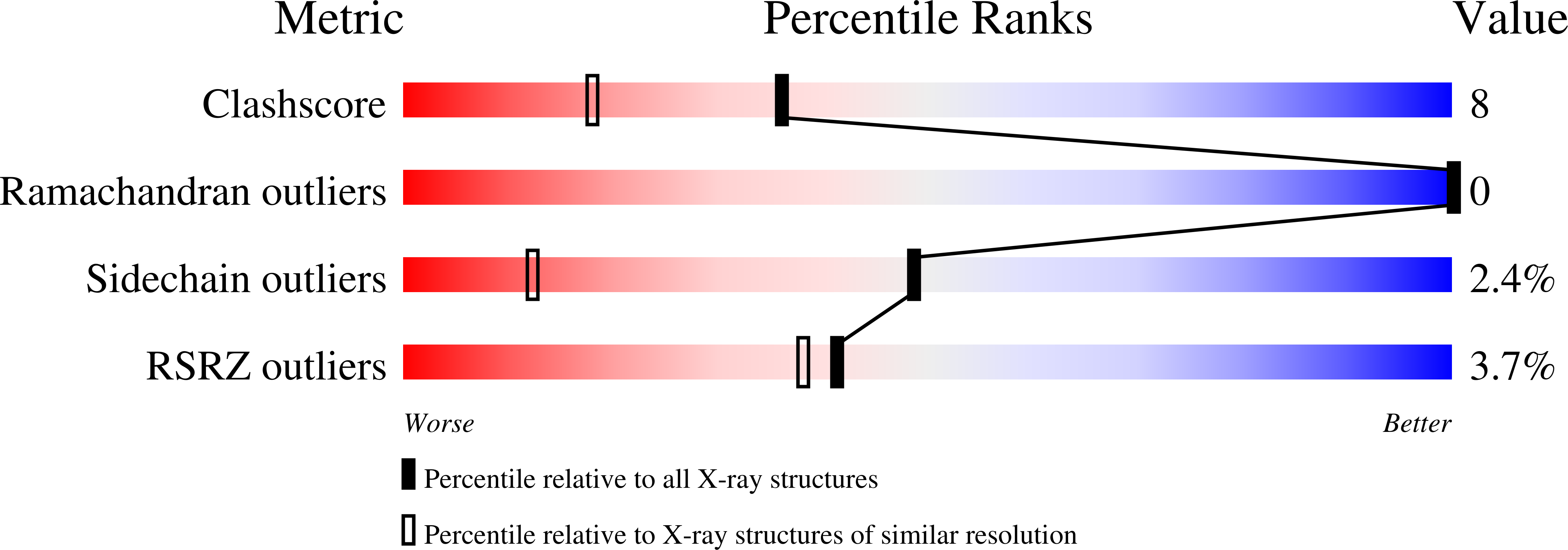Archaeal Protoglobin Structure Indicates New Ligand Diffusion Paths and Modulation of Haem-Reactivity.
Nardini, M., Pesce, A., Thijs, L., Saito, J.A., Dewilde, S., Alam, M., Ascenzi, P., Coletta, M., Ciaccio, C., Moens, L., Bolognesi, M.(2008) EMBO Rep 9: 157
- PubMed: 18188182
- DOI: https://doi.org/10.1038/sj.embor.7401153
- Primary Citation of Related Structures:
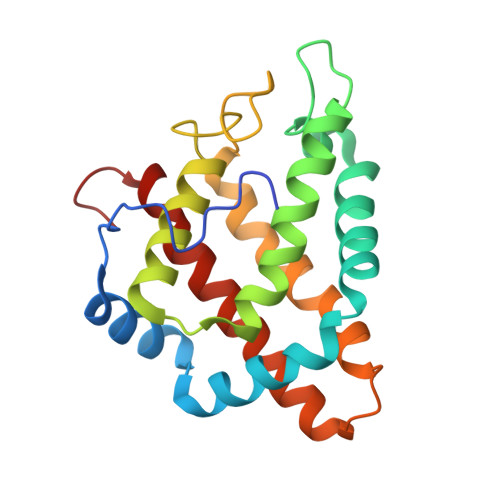
2VEB, 2VEE - PubMed Abstract:
The structural adaptability of the globin fold has been highlighted by the recent discovery of the 2-on-2 haemoglobins, of neuroglobin and cytoglobin. Protoglobin from Methanosarcina acetivorans C2A-a strictly anaerobic methanogenic Archaea-is, to the best of our knowledge, the latest entry adding new variability and functional complexity to the haemoglobin (Hb) superfamily. Here, we report the 1.3 A crystal structure of oxygenated M. acetivorans protoglobin, together with the first insight into its ligand-binding properties. We show that, contrary to all known globins, protoglobin-specific loops and an amino-terminal extension completely bury the haem within the protein matrix. Access of O(2), CO and NO to the haem is granted by the protoglobin-specific apolar tunnels reaching the haem distal site from locations at the B/G and B/E helix interfaces. Functionally, M. acetivorans dimeric protoglobin shows a selectivity ratio for O(2)/CO binding to the haem that favours O(2) ligation and anticooperativity in ligand binding. Both properties are exceptional within the Hb superfamily.
Organizational Affiliation:
Department of Biomolecular Sciences and Biotechnology, CNR-INFM, University of Milano, Via Celoria 26, Milano 20133, Italy.