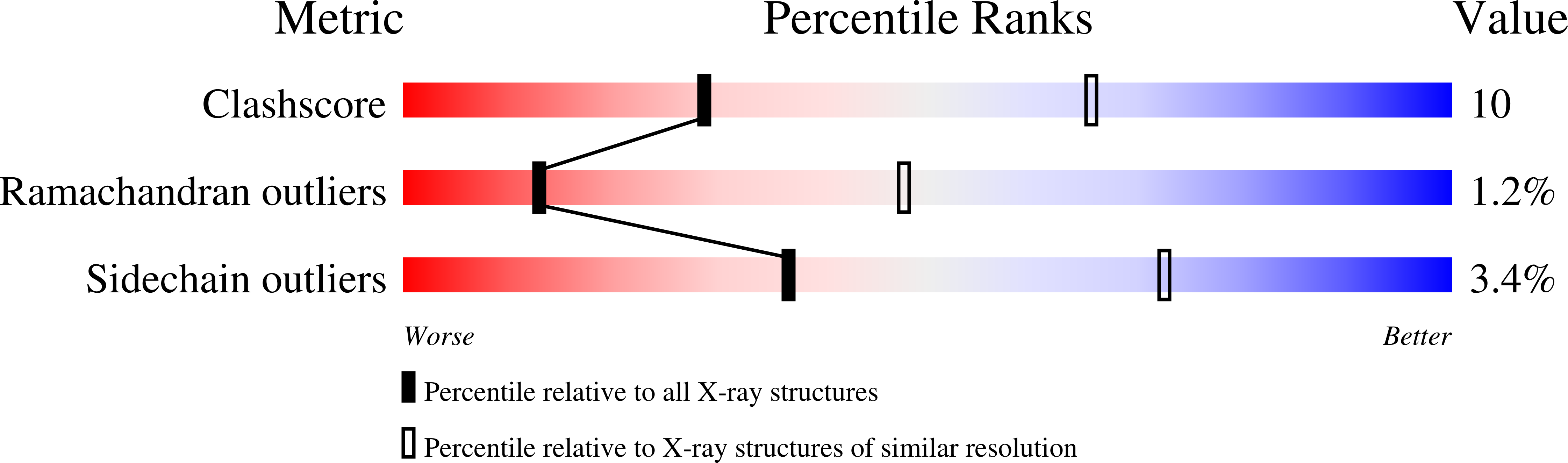
Crystal structures of zinc-free and -bound heme domain of human inducible nitric-oxide synthase. Implications for dimer stability and comparison with endothelial nitric-oxide synthase.
Li, H., Raman, C.S., Glaser, C.B., Blasko, E., Young, T.A., Parkinson, J.F., Whitlow, M., Poulos, T.L.(1999) J Biol Chem 274: 21276-21284
- PubMed: 10409685
- DOI: https://doi.org/10.1074/jbc.274.30.21276
- Primary Citation of Related Structures:
1NSI, 2NSI - PubMed Abstract:
The crystal structures of the heme domain of human inducible nitric-oxide synthase (NOS-2) in zinc-free and -bound states have been solved. In the zinc-free structure, two symmetry-related cysteine residues form a disulfide bond. In the zinc-bound state, these same two cysteine residues form part of a zinc-tetrathiolate (ZnS(4)) center indistinguishable from that observed in the endothelial isoform (NOS-3). As in NOS-3, ZnS(4) plays a key role in stabilizing intersubunit contacts and in maintaining the integrity of the cofactor (tetrahydrobiopterin) binding site of NOS-2. A comparison of NOS-2 and NOS-3 structures illustrates the conservation of quaternary structure, tertiary topology, and substrate and cofactor binding sites, in addition to providing insights on isoform-specific inhibitor design. The structural comparison also reveals that pterin binding does not preferentially stabilize the dimer interface of NOS-2 over NOS-3.
Organizational Affiliation:
Department of Molecular Biology and Biochemistry, University of California at Irvine, Irvine, California 92697, USA.