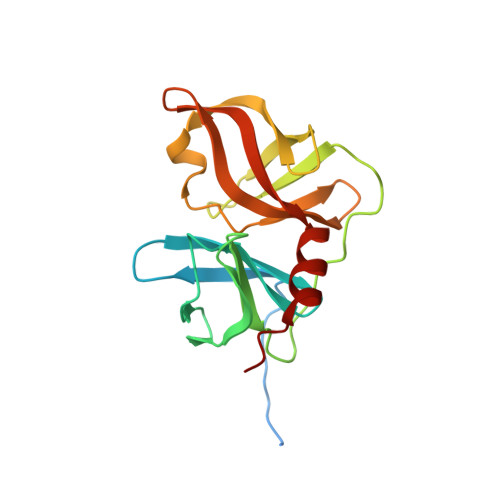
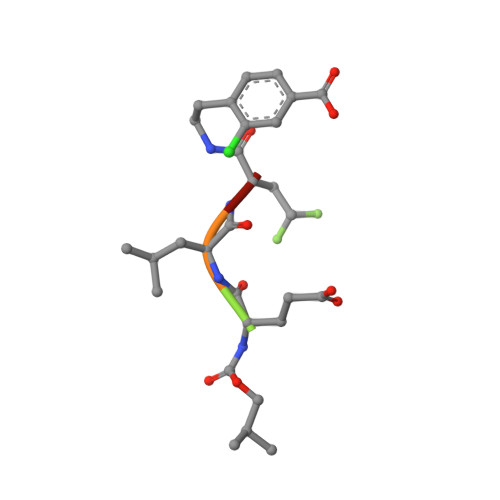
Binding of a noncovalent inhibitor exploiting the S' region stabilizes the hepatitis C virus NS3 protease conformation in the absence of cofactor.
Gallo, M., Pennestri, M., Bottomley, M.J., Barbato, G., Eliseo, T., Paci, M., Narjes, F., De Francesco, R., Summa, V., Koch, U., Bazzo, R., Cicero, D.O.(2009) J Mol Biol 385: 1142-1155
- PubMed: 19061898
- DOI: https://doi.org/10.1016/j.jmb.2008.11.017
- Primary Citation of Related Structures:
2K1Q - PubMed Abstract:
We present the first structure of a noncovalent inhibitor bound to the protease domain of hepatitis C virus NS3 protein (NS3p), solved by NMR. The inhibitor exploits interactions with the S' region of NS3p to form a long-lived complex, although the absence of negative charges strongly reduces the association rate. The inhibitor stabilizes the N-terminal domain of NS3p and the substrate-binding site, and correctly aligns catalytic His-Asp residues. These actions were previously attributed exclusively to the cofactor NS4A, which interacts with the N-terminal domain of the NS3p and functions as an activator in vivo. The structure of the inhibitor/NS3p complex is very similar to that of the NS3p-NS4A complex, showing that binding of the NS4A cofactor is not the only event leading to a stable active-site conformation.
Organizational Affiliation:
Department of Chemical Science and Technology, Università di Roma Tor Vergata Via della Ricerca Scientifica 1, 00133 Rome, Italy.