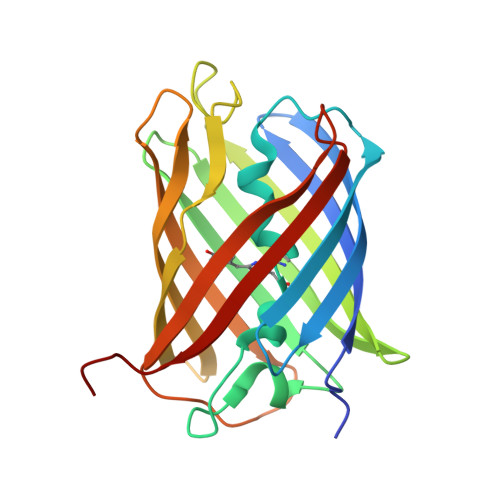
Crystallographic structures of discosoma red fluorescent protein with immature and mature chromophores: linking Peptide bond trans-cis isomerization and acylimine formation in chromophore maturation.
Tubbs, J.L., Tainer, J.A., Getzoff, E.D.(2005) Biochemistry 44: 9833-9840
- PubMed: 16026155
- DOI: https://doi.org/10.1021/bi0472907
- Primary Citation of Related Structures:
1ZGO, 1ZGP, 1ZGQ - PubMed Abstract:
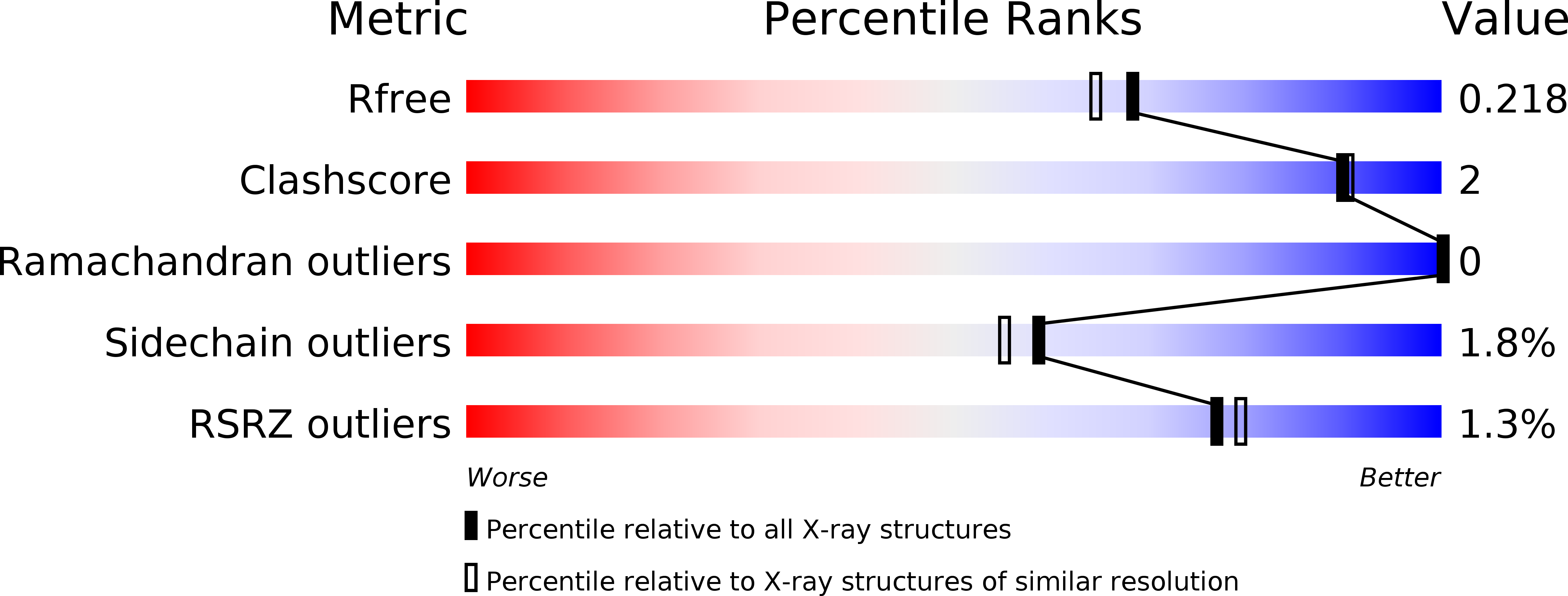
The mature self-synthesizing p-hydroxybenzylideneimidazolinone-like fluorophores of Discosoma red fluorescent protein (DsRed) and Aequorea victoria green fluorescent protein (GFP) are extensively studied as powerful biological markers. Yet, the spontaneous formation of these fluorophores by cyclization, oxidation, and dehydration reactions of tripeptides within their protein environment remains incompletely understood. The mature DsRed fluorophore (Gln 66, Tyr 67, and Gly 68) differs from the GFP fluorophore by an acylimine that results in Gln 66 Calpha planar geometry and by a Phe 65-Gln 66 cis peptide bond. DsRed green-to-red maturation includes a green-fluorescing immature chromophore and requires a chromophore peptide bond trans-cis isomerization that is slow and incomplete. To clarify the unique structural chemistry for the individual immature "green" and mature "red" chromophores of DsRed, we report here the determination and analysis of crystal structures for the wild-type protein (1.4 A resolution), the entirely green DsRed K70M mutant protein (1.9 A resolution), and the DsRed designed mutant Q66M (1.9 A resolution), which shows increased red chromophore relative to the wild-type DsRed. Whereas the mature, red-fluorescing chromophore has the expected cis peptide bond and a sp(2)-hybridized Gln 66 Calpha with planar geometry, the crystal structure of the immature green-fluorescing chromophore of DsRed, presented here for the first time, reveals a trans peptide bond and a sp(3)-hybridized Gln 66 Calpha with tetrahedral geometry. These results characterize a GFP-like immature green DsRed chromophore structure, reveal distinct mature and immature chromophore environments, and furthermore provide evidence for the coupling of acylimine formation with trans-cis isomerization.
Organizational Affiliation:
Department of Molecular Biology and The Skaggs Institute for Chemical Biology, The Scripps Research Institute, La Jolla, California 92037, USA.