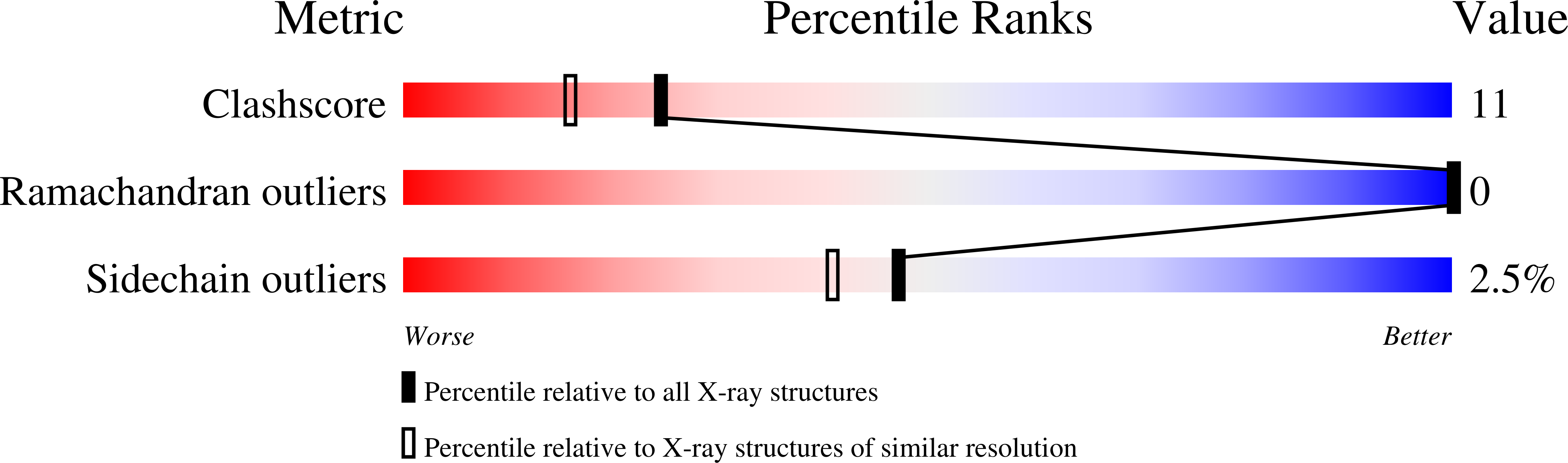1.9 A Resolution Refined Structure of TBP Recognizing the Minor Groove of TATAAAAG
Kim, J.L., Burley, S.K.(1994) Nat Struct Biol 1: 638-653
- PubMed: 7634103
- DOI: https://doi.org/10.1038/nsb0994-638
- Primary Citation of Related Structures:
1VTO - PubMed Abstract:
The three-dimensional structure of a TATA box-binding protein (TBP) from Arabidopsis thaliana complexed with a fourteen base pair oligonucleotide bearing the Adenovirus major late promoter TATA element has been refined at 1.9 A resolution, giving a final crystallographic R-factor of 19.4%. Binding of the monomeric, saddle-shaped alpha/beta protein induces an unprecedented conformational change in the DNA. A detailed structural and functional analysis of this unusual protein-DNA complex is presented, with particular emphasis on the mechanisms of DNA deformation, TATA element recognition, and preinitiation complex assembly.
Organizational Affiliation:
Laboratory of Molecular Biophysics, Howard Hughes Medical Institute, Rockefeller University, New York, NY 10021, USA.