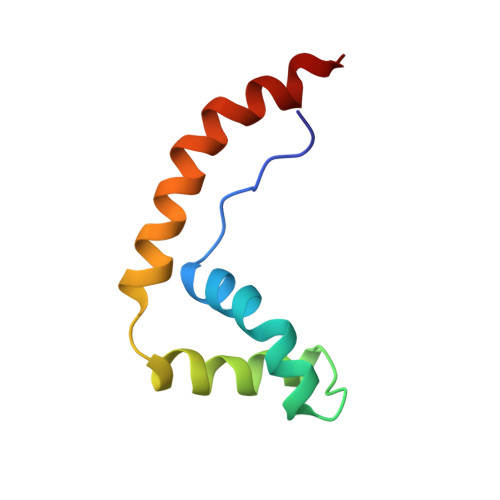
The solution structure and dynamics of the DNA-binding domain of HMG-D from Drosophila melanogaster.
Jones, D.N., Searles, M.A., Shaw, G.L., Churchill, M.E., Ner, S.S., Keeler, J., Travers, A.A., Neuhaus, D.(1994) Structure 2: 609-627
- PubMed: 7922039
- DOI: https://doi.org/10.1016/s0969-2126(00)00063-0
- Primary Citation of Related Structures:
1HMA - PubMed Abstract:
The HMG-box is a conserved DNA-binding motif that has been identified in many high mobility group (HMG) proteins. HMG-D is a non-histone chromosomal protein from Drosophila melanogaster that is closely related to the mammalian HMG-box proteins HMG-1 and HMG-2. Previous structures determined for an HMG-box domain from rat and hamster exhibit the same global topology, but differ significantly in detail. It has been suggested that these differences may arise from hinge motions which allow the protein to adapt to the shape of its target DNA.
Organizational Affiliation:
MRC Laboratory of Molecular Biology, Cambridge, UK.