Hydrogen bonding in human manganese superoxide dismutase containing 3-fluorotyrosine
Ayala, I., Perry, J.J., Szczepanski, J., Tainer, J.A., Vala, M.T., Nick, H.S., Silverman, D.N.(2005) Biophys J 89: 4171-4179
- PubMed: 16150974
- DOI: https://doi.org/10.1529/biophysj.105.060616
- Primary Citation of Related Structures:
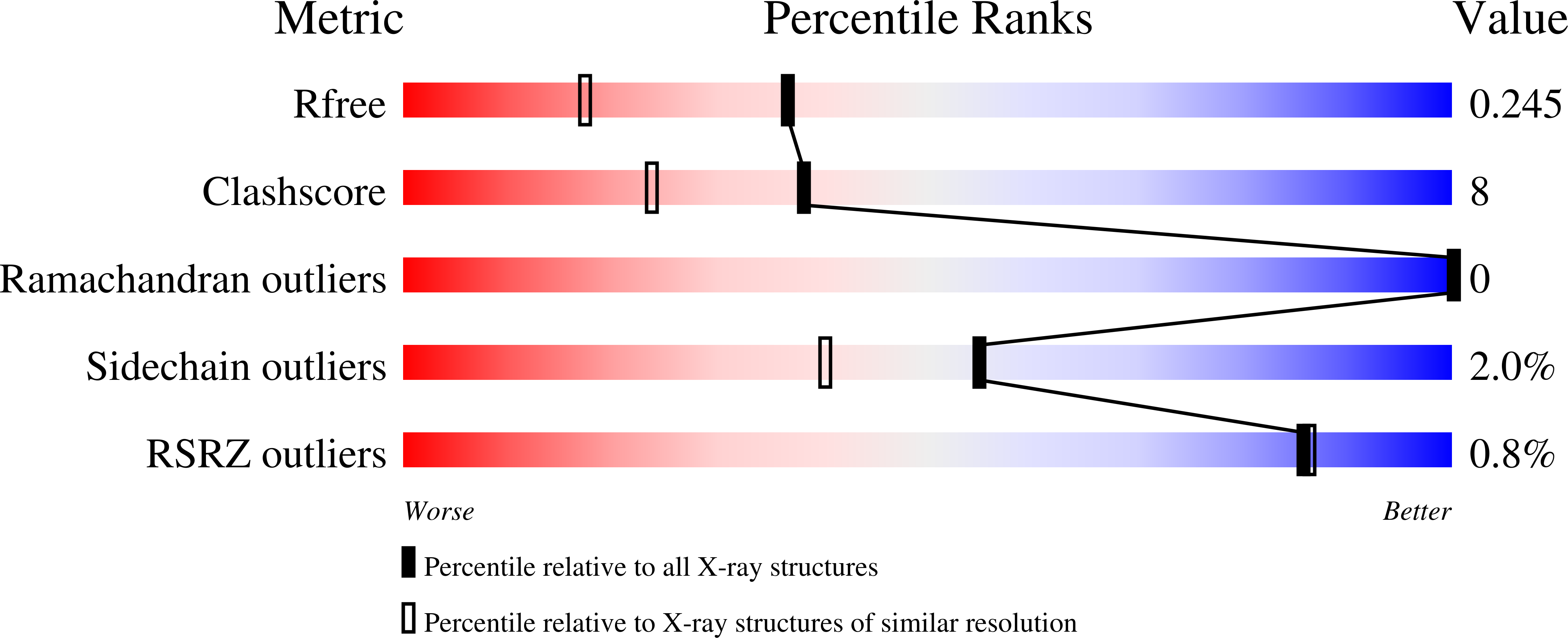
1XDC, 1XIL - PubMed Abstract:
Incorporation of 3-fluorotyrosine and site-specific mutagenesis has been utilized with Fourier transform infrared (FTIR) spectroscopy and x-ray crystallography to elucidate active-site structure and the role of an active-site residue Tyr34 in human manganese superoxide dismutase (MnSOD). Calculated harmonic frequencies at the B3LYP/6-31G** level of theory for L-tyrosine and its 3-fluorine substituted analog are compared to experimental frequencies for vibrational mode assignments. Each of the nine tyrosine residues in each of the four subunits of the homotetramer of human MnSOD was replaced with 3-fluorotyrosine. The crystal structures of the unfluorinated and fluorinated wild-type MnSOD are nearly superimposable with the root mean-square deviation for 198 alpha-carbon atoms at 0.3 A. The FTIR data show distinct vibrational modes arising from 3-fluorotyrosine in MnSOD. Comparison of spectra for wild-type and Y34F MnSOD showed that the phenolic hydroxyl of Tyr34 is hydrogen bonded, acting as a proton donor in the active site. Comparison with crystal structures demonstrates that the hydroxyl of Tyr34 is a hydrogen bond donor to an adjacent water molecule; this confirms the participation of Tyr34 in a network of residues and water molecules that extends from the active site to the adjacent subunit.
Organizational Affiliation:
Department of Pharmacology, University of Florida, Gainesville, Florida 32610, USA.