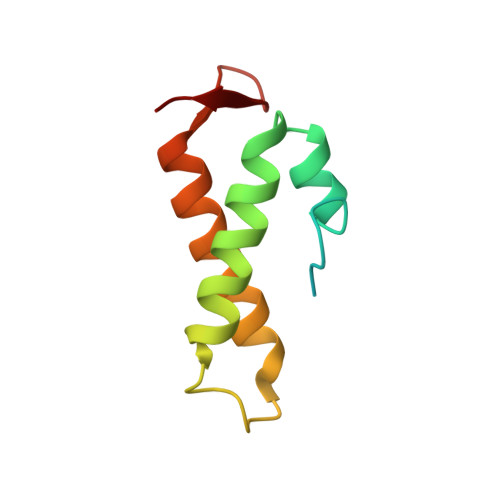
The solution structure of ChaB, a putative membrane ion antiporter regulator from Escherichia coli
Osborne, M.J., Siddiqui, N., Iannuzzi, P., Gehring, K.(2004) BMC Struct Biol 4: 9
- PubMed: 15306028
- DOI: https://doi.org/10.1186/1472-6807-4-9
- Primary Citation of Related Structures:
1SG7 - PubMed Abstract:
ChaB is a putative regulator of ChaA, a Na+/H+ antiporter that also has Ca+/H+ activity in E. coli. ChaB contains a conserved 60-residue region of unknown function found in other bacteria, archaeabacteria and a series of baculoviral proteins. As part of a structural genomics project, the structure of ChaB was elucidated by NMR spectroscopy. The structure of ChaB is composed of 3 alpha-helices and a small sheet that pack tightly to form a fold that is found in the cyclin-box family of proteins. ChaB is distinguished from its putative DNA binding sequence homologues by a highly charged flexible loop region that has weak affinity to Mg2+ and Ca2+ divalent metal ions.
Organizational Affiliation:
Department of Biochemistry, McGill University, 3655 Promenade Sir William Osler, Montreal, Quebec, Canada, H3G 1Y6. mike@bri.nrc.ca