The Kappa-Carrageenase of P. Carrageenovora Features a Tunnel-Shaped Active Site: A Novel Insight in the Evolution of Clan-B Glycoside Hydrolases
Michel, G., Chantalat, L., Duee, E., Barbeyron, T., Henrissat, B., Kloareg, B., Dideberg, O.(2001) Structure 9: 513
- PubMed: 11435116
- DOI: https://doi.org/10.1016/s0969-2126(01)00612-8
- Primary Citation of Related Structures:
1DYP - PubMed Abstract:
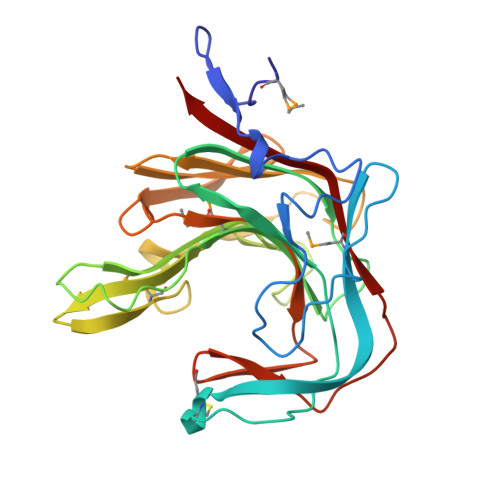
kappa-carrageenans are gel-forming, sulfated 1,3-alpha-1,4-beta-galactans from the cell walls of marine red algae. The kappa-carrageenase from the marine, gram-negative bacterium Pseudoalteromonas carrageenovora degrades kappa-carrageenan both in solution and in solid state by an endoprocessive mechanism. This beta-galactanase belongs to the clan-B of glycoside hydrolases. The structure of P. carrageenovora kappa-carrageenase has been solved to 1.54 A resolution by the multiwavelength anomalous diffraction (MAD) method, using a seleno-methionine-substituted form of the enzyme. The enzyme folds into a curved beta sandwich, with a tunnel-like active site cavity. Another remarkable characteristic is the presence of an arginine residue at subsite -1. The crystal structure of P. carrageenovora kappa-carrageenase is the first three-dimensional structure of a carrageenase. Its tunnel-shaped active site, the first to be reported for enzymes other than cellulases, suggests that such tunnels are associated with the degradation of solid polysaccharides. Clan-B glycoside hydrolases fall into two subgroups, one with catalytic machinery held by an ancestral beta bulge, and the other in which it is held by a regular beta strand. At subsite -1, all of these hydrolases exhibit an aromatic amino acid that interacts with the hexopyranose ring of the monosaccharide undergoing catalysis. In addition, in kappa-carrageenases, an arginine residue recognizes the sulfate-ester substituents of the beta-linked kappa-carrageenan monomers. It also appears that, in addition to the nucleophile and acid/base catalysts, two other amino acids are involved with the catalytic cycle, accelerating the deglycosylation step.
Organizational Affiliation:
Laboratoire de Cristallographie Macromoléculaire, Institut de Biologie Structurale Jean-Pierre Ebel, CNRS/CEA, 41 Avenue des Martyrs, 38027 Cedex 1, Grenoble, France.