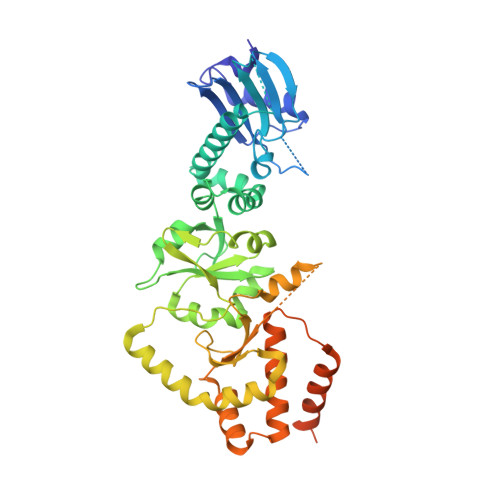
The hexameric structures of human heat shock protein 90
Lee, C.C., Lin, T.W., Ko, T.P., Wang, A.H.-J.(2011) PLoS One 6: e19961-e19961
- PubMed: 21647436
- DOI: https://doi.org/10.1371/journal.pone.0019961
- Primary Citation of Related Structures:
3Q6M, 3Q6N - PubMed Abstract:
The human 90-kDa heat shock protein (HSP90) functions as a dimeric molecular chaperone. HSP90 identified on the cell surface has been found to play a crucial role in cancer invasion and metastasis, and has become a validated anti-cancer target for drug development. It has been shown to self-assemble into oligomers upon heat shock or divalent cations treatment, but the functional role of the oligomeric states in the chaperone cycle is not fully understood. Here we report the crystal structure of a truncated HSP90 that contains the middle segment and the carboxy-terminal domain, termed MC-HSP90. The structure reveals an architecture with triangular bipyramid geometry, in which the building block of the hexameric assembly is a dimer. In solution, MC-HSP90 exists in three major oligomer states, namely dimer, tetramer and hexamer, which were elucidated by size exclusion chromatography and analytical ultracentrifugation. The newly discovered HSP90 isoform HSP90N that lacks the N-terminal ATPase domain also exhibited similar oligomerization states as did MC-HSP90. While lacking the ATPase domain, both MC-HSP90 and HSP90N can self-assemble into a hexameric structure, spontaneously. The crystal structure of MC-HSP90 reveals that, in addition to the C-terminal dimerization domain, the residue W320 in the M domain plays a critical role in its oligomerization. This study not only demonstrates how the human MC-HSP90 forms a hexamer, but also justifies the similar formation of HSP90N by using 3D modeling analysis.
Organizational Affiliation:
Institute of Biological Chemistry, Academia Sinica, Taipei, Taiwan.