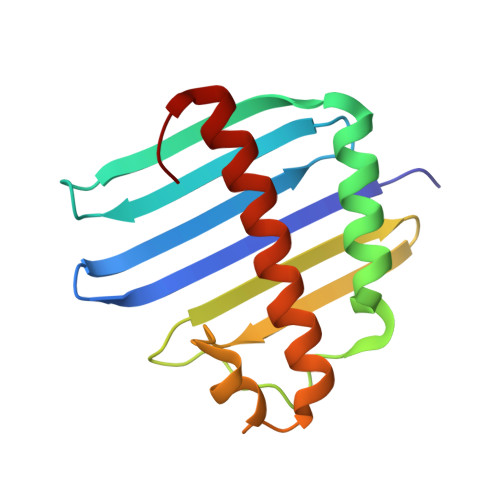
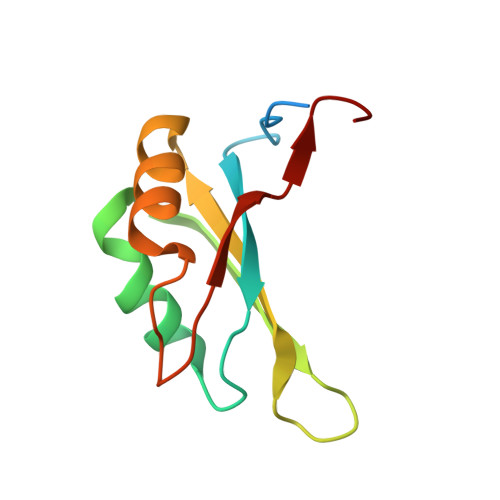
Structure of the y14-magoh core of the exon junction complex.
Lau, C.K., Diem, M.D., Dreyfuss, G., Van Duyne, G.D.(2003) Curr Biol 13: 933-941
- PubMed: 12781131
- DOI: https://doi.org/10.1016/s0960-9822(03)00328-2
- Primary Citation of Related Structures:
1P27 - PubMed Abstract:
Splicing of pre-mRNA in eukaryotes imprints the resulting mRNA with a specific multiprotein complex, the exon-exon junction complex (EJC), at the sites of intron removal. The proteins of the EJC, Y14, Magoh, Aly/REF, RNPS1, Srm160, and Upf3, play critical roles in postsplicing processing, including nuclear export and cytoplasmic localization of the mRNA, and the nonsense-mediated mRNA decay (NMD) surveillance process. Y14 and Magoh are of particular interest because they remain associated with the mRNA in the same position after its export to the cytoplasm and require translation of the mRNA for removal. This tenacious, persistent, splicing-dependent, yet RNA sequence-independent, association suggests an important signaling function and must require distinct structural features for these proteins.
Organizational Affiliation:
Howard Hughes Medical Institute and Department of Biochemistry and Biophysics, University of Pennsylvania School of Medicine, Philadelphia, PA 19104-6059, USA.