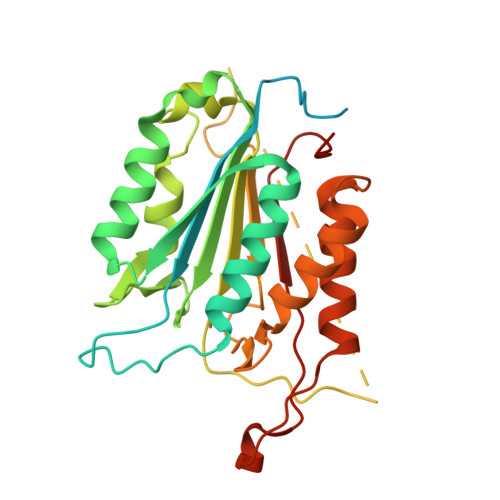
The structures of caspases-1, -3, -7 and -8 reveal the basis for substrate and inhibitor selectivity.
Wei, Y., Fox, T., Chambers, S.P., Sintchak, J., Coll, J.T., Golec, J.M., Swenson, L., Wilson, K.P., Charifson, P.S.(2000) Chem Biol 7: 423-432
- PubMed: 10873833
- DOI: https://doi.org/10.1016/s1074-5521(00)00123-x
- Primary Citation of Related Structures:
1F1J - PubMed Abstract:
Peptide inhibitors of caspases have helped define the role of these cysteine proteases in biology. Structural and biochemical characterization of the caspase enzymes may contribute to the development of new drugs for the treatment of caspase-mediated inflammation and apoptosis.
Organizational Affiliation:
Vertex Pharmaceuticals, Cambridge, MA 02139-4242, USA.