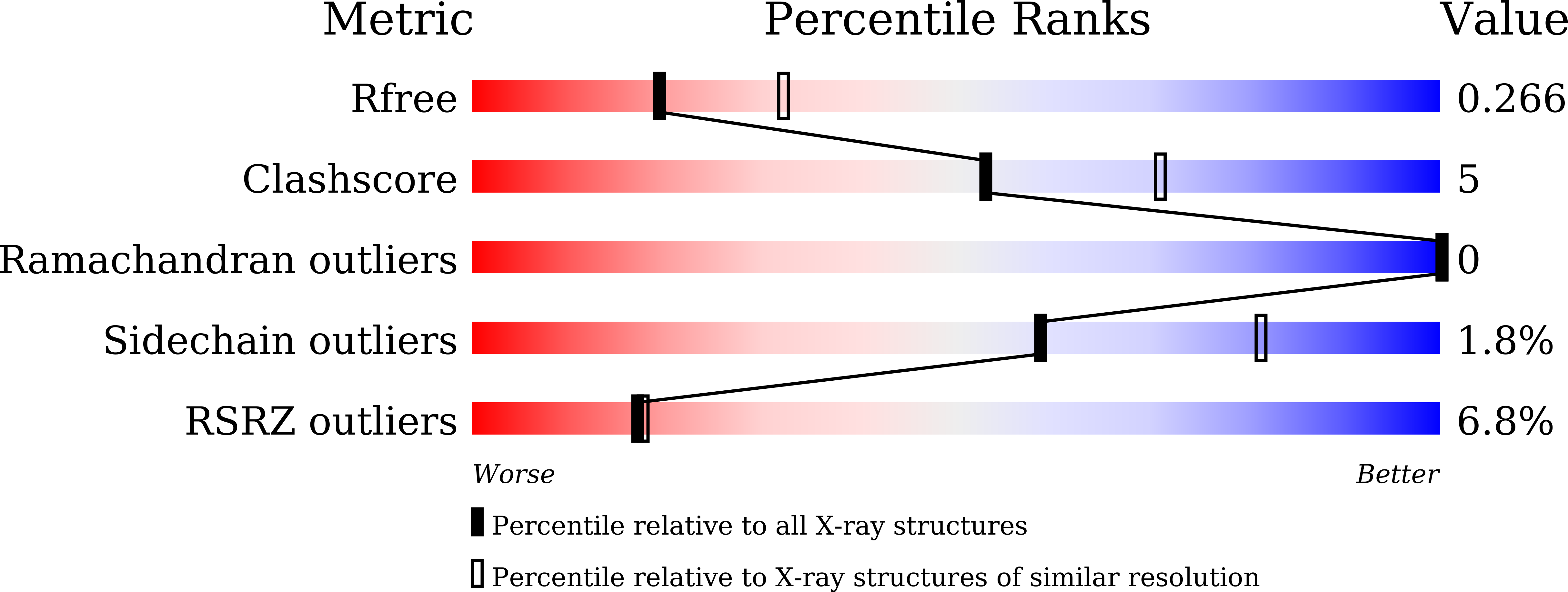
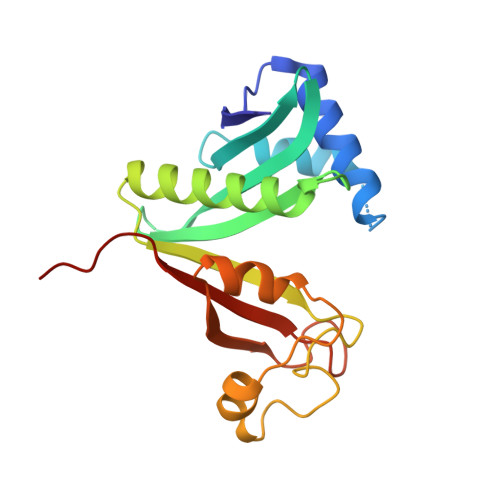
Structural analysis of a novel substrate-free form of the aminoglycoside 6'-N-acetyltransferase from Enterococcus faecium.
Jang, H., Kwon, S., Jeong, C.S., Lee, C.W., Hwang, J., Jung, K.H., Lee, J.H., Park, H.H.(2020) Acta Crystallogr F Struct Biol Commun 76: 364-371
- PubMed: 32744248
- DOI: https://doi.org/10.1107/S2053230X20009735
- Primary Citation of Related Structures:
7BXZ - PubMed Abstract:
Aminoglycoside acetyltransferases (AACs) catalyze the transfer of an acetyl group between acetyl-CoA and an aminoglycoside, producing CoA and an acetylated aminoglycoside. AAC(6')-Ii enzymes target the amino group linked to the 6' C atom in an aminoglycoside. Several structures of the AAC(6')-Ii from Enterococcus faecium [Ef-AAC(6')-Ii] have been reported to date. However, the detailed mechanism of its enzymatic function remains elusive. In this study, the crystal structure of Ef-AAC(6')-Ii was determined in a novel substrate-free form. Based on structural analysis, it is proposed that Ef-AAC(6')-Ii sequentially undergoes conformational selection and induced fit for substrate binding. These results therefore provide a novel viewpoint on the mechanism of action of Ef-AAC(6')-Ii.
Organizational Affiliation:
College of Pharmacy, Chung-Ang University, Dongjak-gu, Seoul 06974, Republic of Korea.