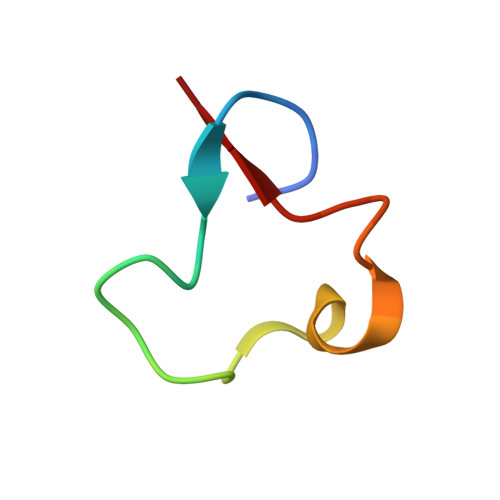
Discovery of Ubonodin, an Antimicrobial Lasso Peptide Active against Members of the Burkholderia cepacia Complex.
Cheung-Lee, W.L., Parry, M.E., Zong, C., Cartagena, A.J., Darst, S.A., Connell, N.D., Russo, R., Link, A.J.(2020) Chembiochem 21: 1335-1340
- PubMed: 31765515
- DOI: https://doi.org/10.1002/cbic.201900707
- Primary Citation of Related Structures:
6POR - PubMed Abstract:
We report the heterologous expression, structure, and antimicrobial activity of a lasso peptide, ubonodin, encoded in the genome of Burkholderia ubonensis. The topology of ubonodin is unprecedented amongst lasso peptides, with 18 of its 28 amino acids found in the mechanically bonded loop segment. Ubonodin inhibits RNA polymerase in vitro and has potent antimicrobial activity against several pathogenic members of the Burkholderia genus, most notably B. cepacia and B. multivorans, causative agents of lung infections in cystic fibrosis patients.
Organizational Affiliation:
Department of Chemical and Biological Engineering, Princeton University, 207 Hoyt Laboratory, Princeton, NJ, 08544, USA.