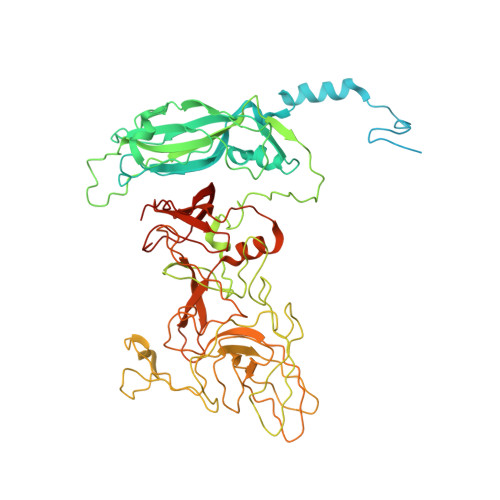
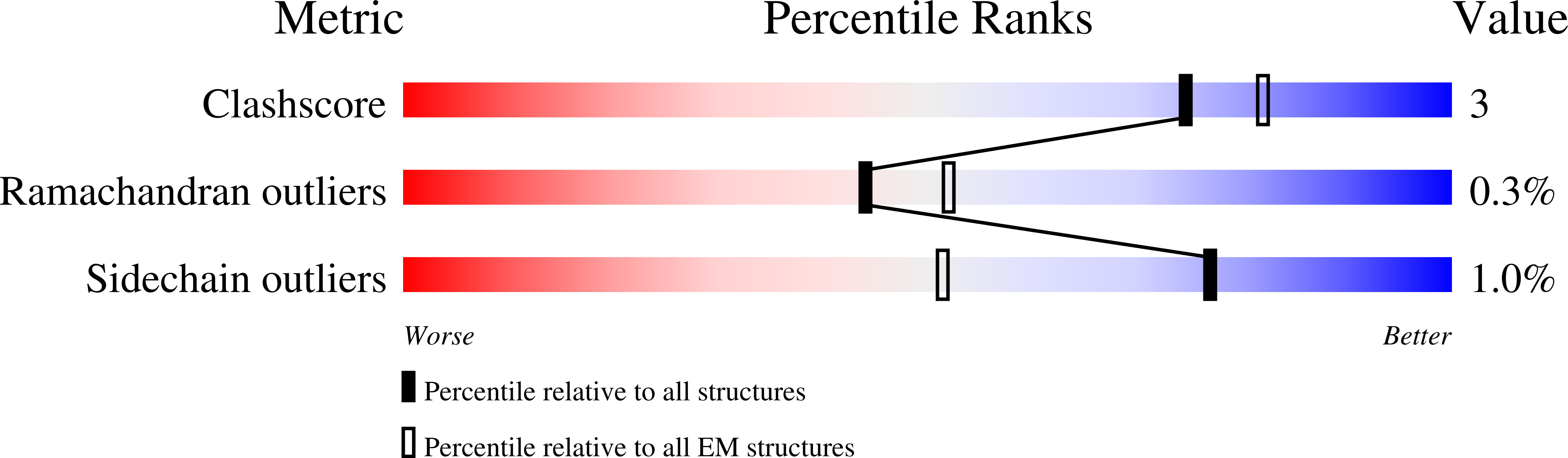Calicivirus VP2 forms a portal-like assembly following receptor engagement.
Conley, M.J., McElwee, M., Azmi, L., Gabrielsen, M., Byron, O., Goodfellow, I.G., Bhella, D.(2019) Nature 565: 377-381
- PubMed: 30626974
- DOI: https://doi.org/10.1038/s41586-018-0852-1
- Primary Citation of Related Structures:
6GSH, 6GSI - PubMed Abstract:
To initiate infection, many viruses enter their host cells by triggering endocytosis following receptor engagement. However, the mechanisms by which non-enveloped viruses escape the endosome are poorly understood. Here we present near-atomic-resolution cryo-electron microscopy structures for feline calicivirus both undecorated and labelled with a soluble fragment of its cellular receptor, feline junctional adhesion molecule A. We show that VP2, a minor capsid protein encoded by all caliciviruses 1,2 , forms a large portal-like assembly at a unique three-fold axis of symmetry, following receptor engagement. This assembly-which was not detected in undecorated virions-is formed of twelve copies of VP2, arranged with their hydrophobic N termini pointing away from the virion surface. Local rearrangement at the portal site leads to the opening of a pore in the capsid shell. We hypothesize that the portal-like assembly functions as a channel for the delivery of the calicivirus genome, through the endosomal membrane, into the cytoplasm of a host cell, thereby initiating infection. VP2 was previously known to be critical for the production of infectious virus 3 ; our findings provide insights into its structure and function that advance our understanding of the Caliciviridae.
Organizational Affiliation:
Medical Research Council University of Glasgow Centre for Virus Research, Glasgow, UK.