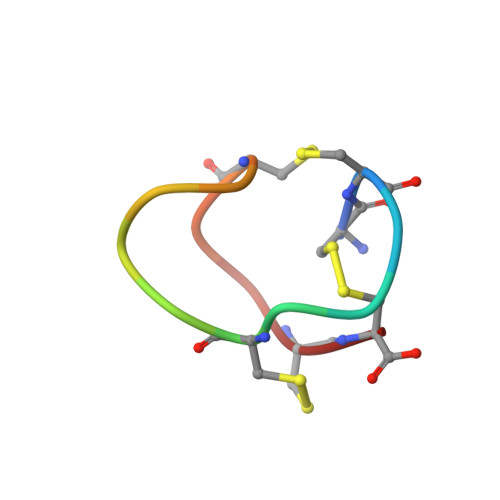
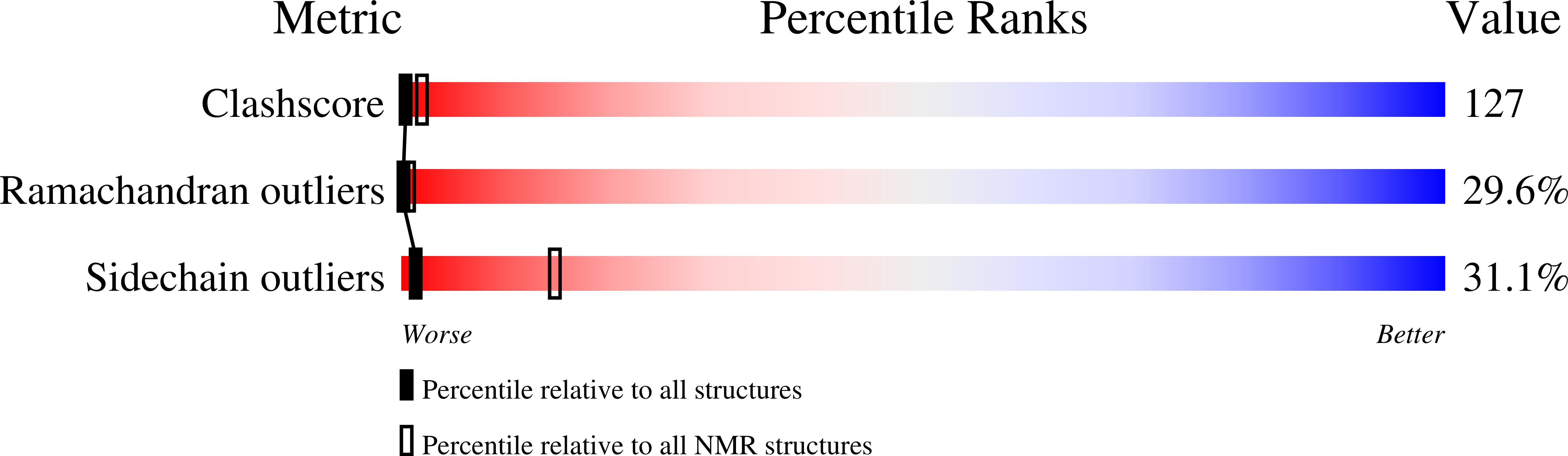Structural plasticity of mini-M conotoxins - expression of all mini-M subtypes by Conus regius.
Franco, A., Dovell, S., Moller, C., Grandal, M., Clark, E., Mari, F.(2018) FEBS J 285: 887-902
- PubMed: 29283511
- DOI: https://doi.org/10.1111/febs.14372
- Primary Citation of Related Structures:
6BX9 - PubMed Abstract:
The mini-M conotoxins are peptidic scaffolds found in the venom of cones snails. These scaffolds are tightly folded structures held together by three disulfide bonds with a CC-C-C-CC arrangement (conotoxin framework III) and belong to the M Superfamily of conotoxins. Here, we describe mini-M conotoxins from the venom of Conus regius, a Western Atlantic worm-hunting cone snail species using transcriptomic and peptidomic analyses. These C. regius conotoxins belong to three different subtypes: M1, M2, and M3. The subtypes show little sequence homology, and their loop sizes (intercysteine amino acid chains) vary significantly. The mini-Ms isolated from dissected venom contains preferentially hydroxylated proline residues, thus augmenting the structural reach of this conotoxin class. Using 2D-NMR methods, we have determined the 3D structure of reg3b, an M2 subtype conotoxin, which shows a constrained multi-turn scaffold. The structural diversity found within mini-M conotoxin scaffolds of C. regius is indicative of structural hypervariability of the conotoxin M superfamily that is not seen in other superfamilies. These stable minimalistic scaffolds may be investigated for the development of engineered peptides for therapeutic applications.
Organizational Affiliation:
Department of Chemistry and Biochemistry, Florida Atlantic University, Boca Raton, FL, USA.