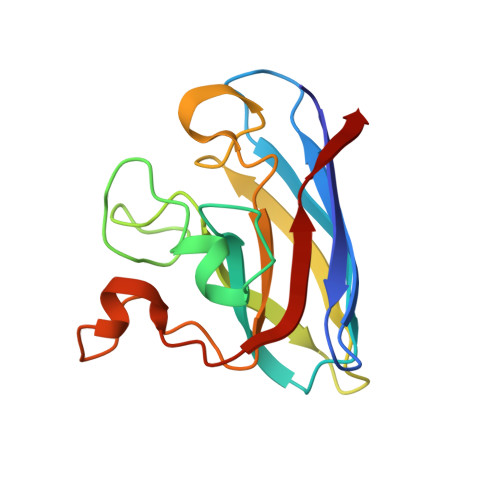
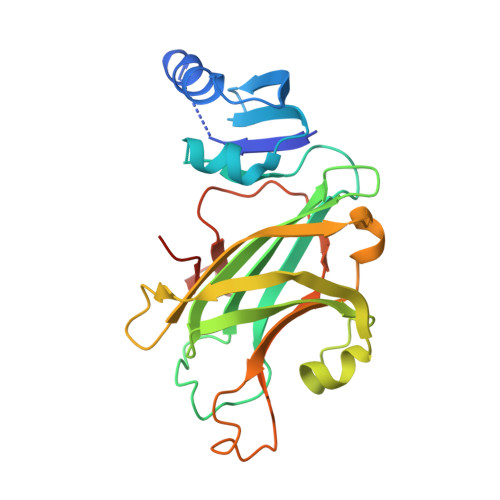
Copper-zinc superoxide dismutase is activated through a sulfenic acid intermediate at a copper ion entry site.
Fetherolf, M.M., Boyd, S.D., Taylor, A.B., Kim, H.J., Wohlschlegel, J.A., Blackburn, N.J., Hart, P.J., Winge, D.R., Winkler, D.D.(2017) J Biol Chem 292: 12025-12040
- PubMed: 28533431
- DOI: https://doi.org/10.1074/jbc.M117.775981
- Primary Citation of Related Structures:
5U9M - PubMed Abstract:
Metallochaperones are a diverse family of trafficking molecules that provide metal ions to protein targets for use as cofactors. The copper chaperone for superoxide dismutase (Ccs1) activates immature copper-zinc superoxide dismutase (Sod1) by delivering copper and facilitating the oxidation of the Sod1 intramolecular disulfide bond. Here, we present structural, spectroscopic, and cell-based data supporting a novel copper-induced mechanism for Sod1 activation. Ccs1 binding exposes an electropositive cavity and proposed "entry site" for copper ion delivery on immature Sod1. Copper-mediated sulfenylation leads to a sulfenic acid intermediate that eventually resolves to form the Sod1 disulfide bond with concomitant release of copper into the Sod1 active site. Sod1 is the predominant disulfide bond-requiring enzyme in the cytoplasm, and this copper-induced mechanism of disulfide bond formation obviates the need for a thiol/disulfide oxidoreductase in that compartment.
Organizational Affiliation:
Department of Medicine, University of Utah Health Sciences Center School of Medicine, Salt Lake City, Utah 84132-2408; Department of Biochemistry, University of Utah, Salt Lake City, Utah 84112-5650.