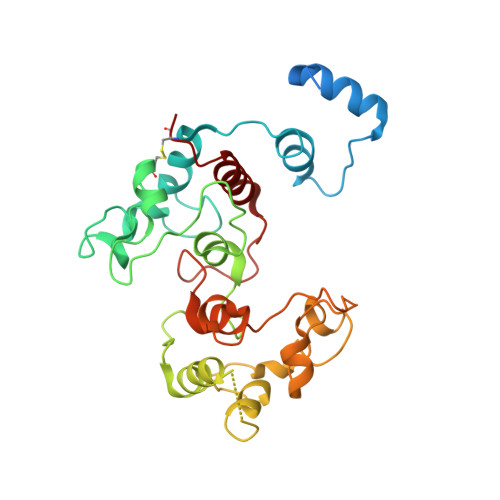
Crystallization and preliminary crystallographic studies of FoxE from Rhodobacter ferrooxidans SW2, an Fe(II) oxidoreductase involved in photoferrotrophy.
Pereira, L., Saraiva, I.H., Coelho, R., Newman, D.K., Louro, R.O., Frazao, C.(2012) Acta Crystallogr Sect F Struct Biol Cryst Commun 68: 1106-1108
- PubMed: 22949206
- DOI: https://doi.org/10.1107/S174430911203271X
- Primary Citation of Related Structures:
5MVO - PubMed Abstract:
FoxE is a protein encoded by the foxEYZ operon of Rhodobacter ferrooxidans SW2 that is involved in Fe(II)-based anoxygenic photosynthesis (`photoferrotrophy'). It is thought to reside in the periplasm, where it stimulates light-dependent Fe(II) oxidation. It contains 259 residues, including two haem c-binding motifs. As no three-dimensional model is available and there is no structure with a similar sequence, crystals of FoxE were produced. They diffracted to 2.44 Å resolution using synchrotron radiation at the Fe edge. The phase problem was solved by SAD using SHELXC/D/E and the experimental maps confirmed the presence of two haems per molecule.
Organizational Affiliation:
Instituto de Tecnologia Química e Biológica, Universidade Nova de Lisboa, Apartado 127, 2781-901 Oeiras, Portugal.