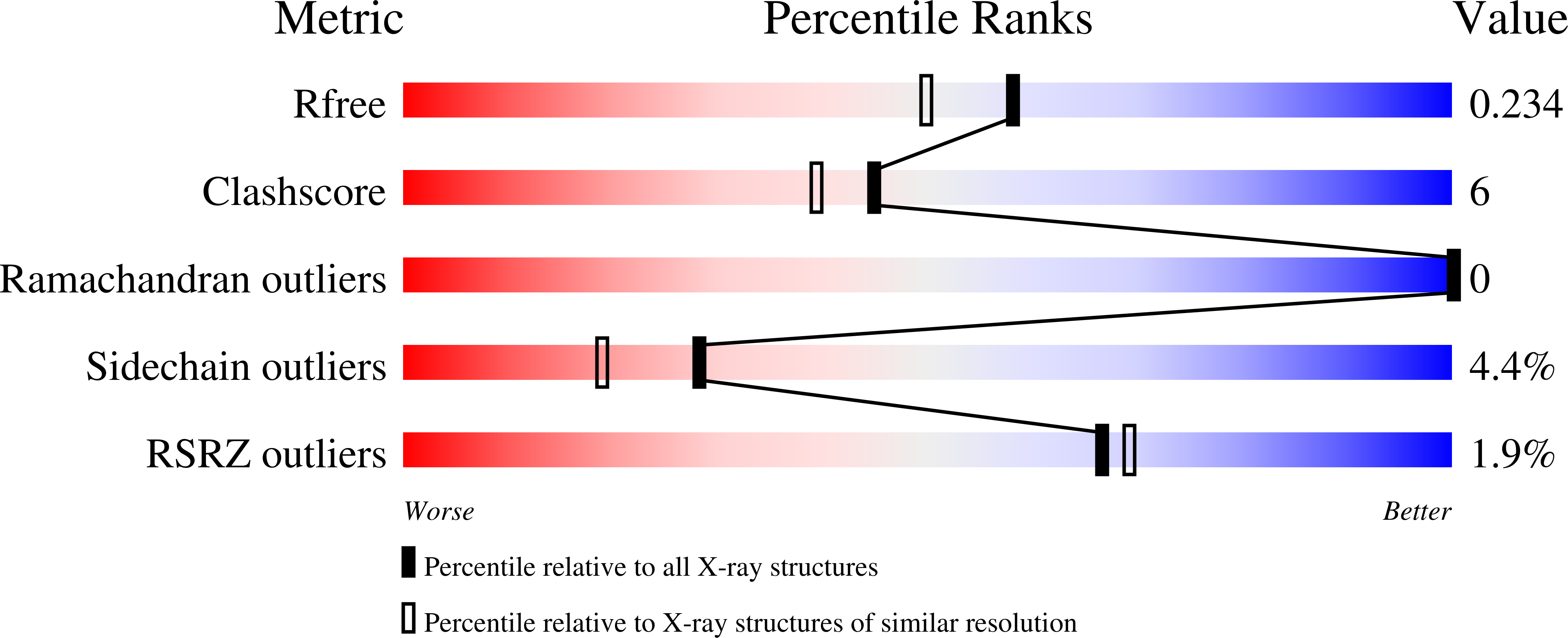
Identification of potent, selective KDM5 inhibitors.
Gehling, V.S., Bellon, S.F., Harmange, J.C., LeBlanc, Y., Poy, F., Odate, S., Buker, S., Lan, F., Arora, S., Williamson, K.E., Sandy, P., Cummings, R.T., Bailey, C.M., Bergeron, L., Mao, W., Gustafson, A., Liu, Y., VanderPorten, E., Audia, J.E., Trojer, P., Albrecht, B.K.(2016) Bioorg Med Chem Lett 26: 4350-4354
- PubMed: 27476424
- DOI: https://doi.org/10.1016/j.bmcl.2016.07.026
- Primary Citation of Related Structures:
5KR7 - PubMed Abstract:
This communication describes the identification and optimization of a series of pan-KDM5 inhibitors derived from compound 1, a hit initially identified against KDM4C. Compound 1 was optimized to afford compound 20, a 10nM inhibitor of KDM5A. Compound 20 is highly selective for the KDM5 enzymes versus other histone lysine demethylases and demonstrates activity in a cellular assay measuring the increase in global histone 3 lysine 4 tri-methylation (H3K4me3). In addition compound 20 has good ADME properties, excellent mouse PK, and is a suitable starting point for further optimization.
Organizational Affiliation:
Constellation Pharmaceuticals, Inc., 215 First Street, Suite 200, Cambridge, MA 02142, United States. Electronic address: victor.gehling@constellationpharma.com.