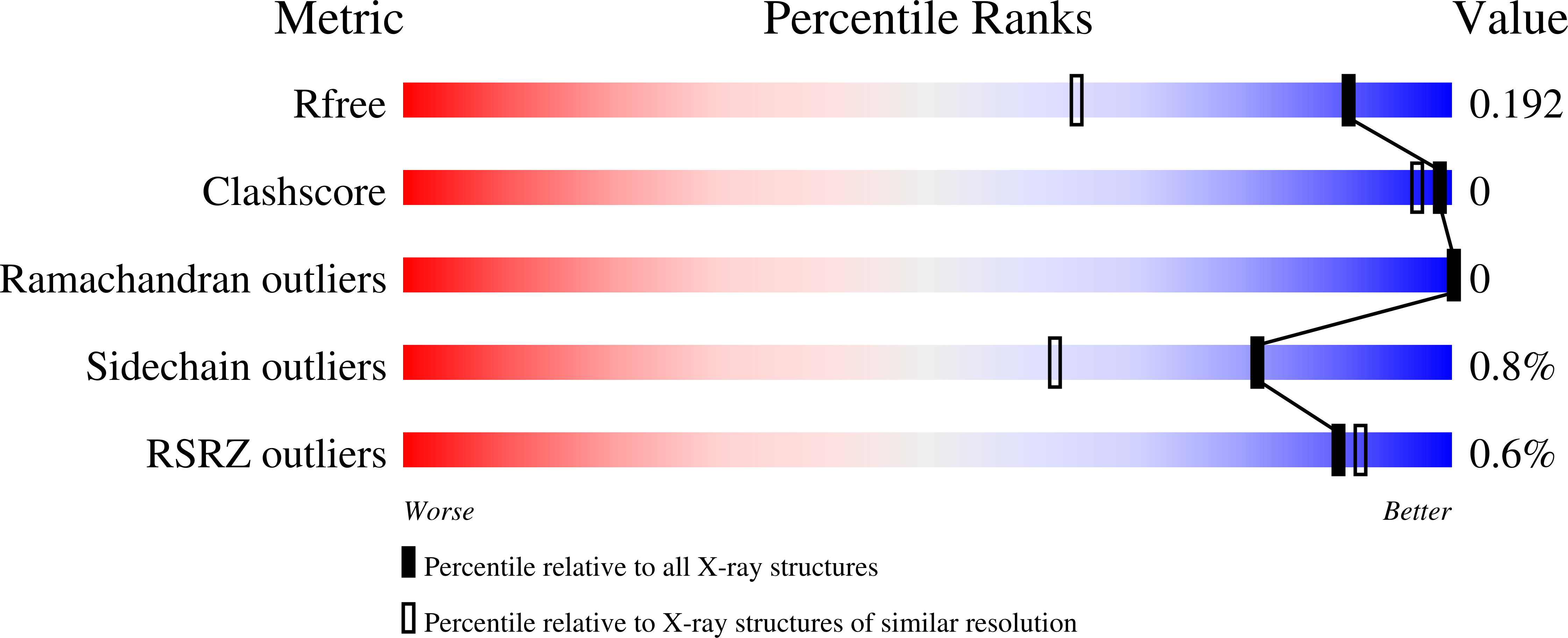
High-resolution crystal structures of two crystal forms of human cyclophilin D in complex with PEG 400 molecules.
Valasani, K.R., Carlson, E.A., Battaile, K.P., Bisson, A., Wang, C., Lovell, S., ShiDu Yan, S.(2014) Acta Crystallogr Sect F Struct Biol Cryst Commun 70: 717-722
- PubMed: 24915078
- DOI: https://doi.org/10.1107/S2053230X14009480
- Primary Citation of Related Structures:
4O8H, 4O8I - PubMed Abstract:
Cyclophilin D (CypD) is a key mitochondrial target for amyloid-β-induced mitochondrial and synaptic dysfunction and is considered a potential drug target for Alzheimer's disease. The high-resolution crystal structures of primitive orthorhombic (CypD-o) and primitive tetragonal (CypD-t) forms have been determined to 1.45 and 0.85 Å resolution, respectively, and are nearly identical structurally. Although an isomorphous structure of CypD-t has previously been reported, the structure reported here was determined at atomic resolution, while CypD-o represents a new crystal form for this protein. In addition, each crystal form contains a PEG 400 molecule bound to the same region along with a second PEG 400 site in CypD-t which occupies the cyclosporine A inhibitor binding site of CypD. Highly precise structural information for CypD should be extremely useful for discerning the detailed interaction of small molecules, particularly drugs and/or inhibitors, bound to CypD. The 0.85 Å resolution structure of CypD-t is the highest to date for any CypD structure.
Organizational Affiliation:
Department of Pharmacology and Toxicology and Higuchi Bioscience Center, University of Kansas, Lawrence, KS 66047, USA.