Crystal Structure of GH18 Chitinase from Cycad, Cycas revoluta
Umemoto, N., Numata, T., Ohnuma, T., Osawa, T., Taira, T., Fukamizo, T.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Chitinase A | 348 | Cycas revoluta | Mutation(s): 0 Gene Names: crchiA EC: 3.2.1.14 | 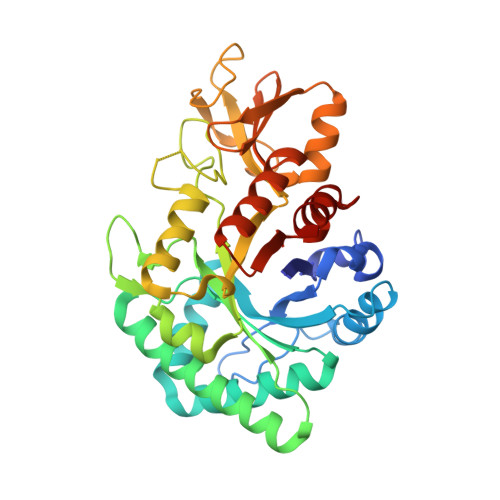 | |
UniProt | |||||
Find proteins for Q4W6L6 (Cycas revoluta) Explore Q4W6L6 Go to UniProtKB: Q4W6L6 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q4W6L6 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| B3P Query on B3P | E [auth A] | 2-[3-(2-HYDROXY-1,1-DIHYDROXYMETHYL-ETHYLAMINO)-PROPYLAMINO]-2-HYDROXYMETHYL-PROPANE-1,3-DIOL C11 H26 N2 O6 HHKZCCWKTZRCCL-UHFFFAOYSA-N |  | ||
| ACT Query on ACT | D [auth A] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_900017 Query on PRD_900017 | B | triacetyl-beta-chitotriose | Oligosaccharide / Inhibitor |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 57.822 | α = 90 |
| b = 64.036 | β = 90 |
| c = 85.986 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| MOLREP | phasing |
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |