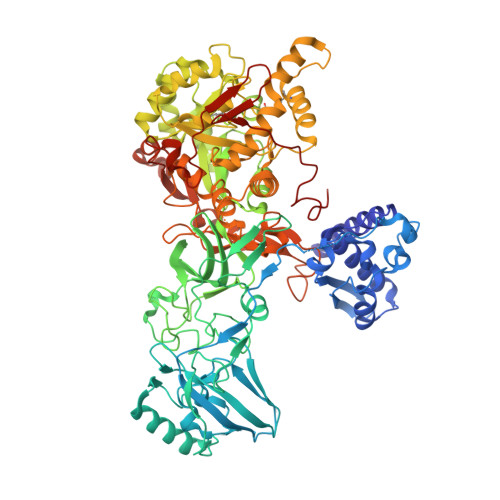
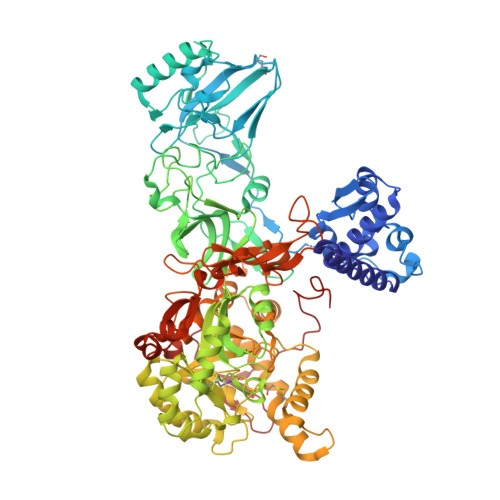
Structural and functional studies on urease from pigeon pea (Cajanus cajan)
Balasubramanian, A., Durairajpandian, V., Elumalai, S., Mathivanan, N., Munirajan, A.K., Ponnuraj, K.(2013) Int J Biol Macromol 58C: 301-309
- PubMed: 23624166
- DOI: https://doi.org/10.1016/j.ijbiomac.2013.04.055
- Primary Citation of Related Structures:
4G7E - PubMed Abstract:
Urease is an enzyme that catalyzes the hydrolysis of urea, forming ammonia and carbon dioxide, and is found in plants, microorganisms and invertebrates. Although plant and bacterial ureases are closely related at amino acid and at the structural level, the insecticidal activity is seen only in the plant ureases. In contrast, both plant and bacterial ureases exhibit antifungal activity. These two biological properties are independent of its ureolytic activity. However, till date the mechanism(s) behind the insecticidal and fungicidal activity of ureases are not clearly understood. Here we report the crystal structure of pigeon pea urease (PPU, Cajanus cajan) which is the second structure from the plant source. We have deduced the amino acid sequence of PPU and also report here studies on its stability, insecticidal and antifungal activity. PPU exhibits cellulase activity. Based on the structural analysis of PPU and docking studies with cellopentoase we propose a possible mechanism of antifungal activity of urease.
Organizational Affiliation:
Centre of Advanced Study in Crystallography and Biophysics University of Madras, Guindy Campus, Chennai 600025, India.