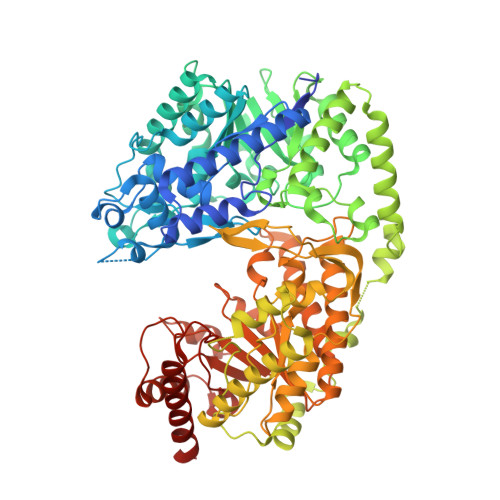
Structure of Candida albicans methionine synthase determined by employing surface residue mutagenesis.
Ubhi, D., Kavanagh, K.L., Monzingo, A.F., Robertus, J.D.(2011) Arch Biochem Biophys 513: 19-26
- PubMed: 21689631
- DOI: https://doi.org/10.1016/j.abb.2011.06.002
- Primary Citation of Related Structures:
3PPC, 3PPF, 3PPG, 3PPH - PubMed Abstract:
Fungal methionine synthase, Met6p, transfers a methyl group from 5-methyl-tetrahydrofolate to homocysteine to generate methionine. The enzyme is essential to fungal growth and is a potential anti-fungal drug design target. We have characterized the enzyme from the pathogen Candida albicans but were unable to crystallize it in native form. We converted Lys103, Lys104, and Glu107 all to Tyr (Met6pY), Thr (Met6pT) and Ala (Met6pA). All variants showed wild-type kinetic activity and formed useful crystals, each with unique crystal packing. In each case the mutated residues participated in beneficial crystal contacts. We have solved the three structures at 2.0-2.8Å resolution and analyzed crystal packing, active-site residues, and similarity to other known methionine synthase structures. C. albicans Met6p has a two domain structure with each of the domains having a (βα)(8)-barrel fold. The barrels are arranged face-to-face and the active site is located in a cleft between the two domains. Met6p utilizes a zinc ion for catalysis that is bound in the C-terminal domain and ligated by four conserved residues: His657, Cys659, Glu679 and Cys739.
Organizational Affiliation:
Institute for Cellular and Molecular Biology, Department of Chemistry and Biochemistry, University of Texas at Austin, United States.