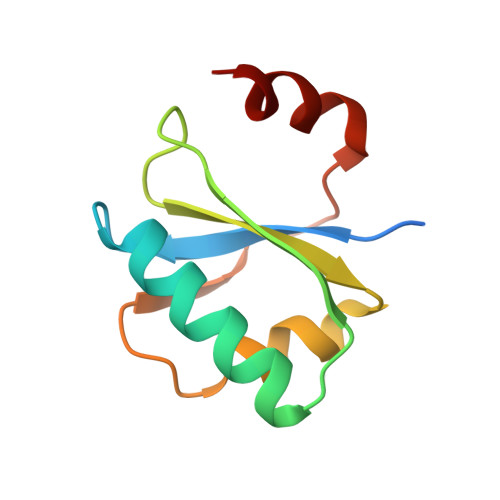
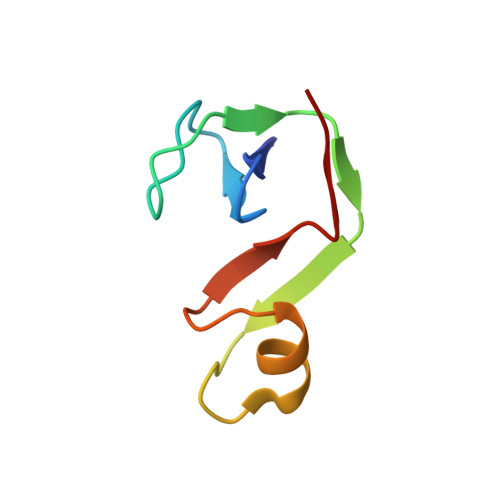
Spt4/5 stimulates transcription elongation through the RNA polymerase clamp coiled-coil motif.
Hirtreiter, A., Damsma, G.E., Cheung, A.C., Klose, D., Grohmann, D., Vojnic, E., Martin, A.C., Cramer, P., Werner, F.(2010) Nucleic Acids Res 38: 4040-4051
- PubMed: 20197319
- DOI: https://doi.org/10.1093/nar/gkq135
- Primary Citation of Related Structures:
3LPE - PubMed Abstract:
Spt5 is the only known RNA polymerase-associated factor that is conserved in all three domains of life. We have solved the structure of the Methanococcus jannaschii Spt4/5 complex by X-ray crystallography, and characterized its function and interaction with the archaeal RNAP in a wholly recombinant in vitro transcription system. Archaeal Spt4 and Spt5 form a stable complex that associates with RNAP independently of the DNA-RNA scaffold of the elongation complex. The association of Spt4/5 with RNAP results in a stimulation of transcription processivity, both in the absence and the presence of the non-template strand. A domain deletion analysis reveals the molecular anatomy of Spt4/5--the Spt5 Nus-G N-terminal (NGN) domain is the effector domain of the complex that both mediates the interaction with RNAP and is essential for its elongation activity. Using a mutagenesis approach, we have identified a hydrophobic pocket on the Spt5 NGN domain as binding site for RNAP, and reciprocally the RNAP clamp coiled-coil motif as binding site for Spt4/5.
Organizational Affiliation:
Division of Biosciences, University College London, Institute for Structural and Molecular Biology, Darwin Building, Gower Street, London WC1E 6BT, UK.