Structural characterization of a family of cytochromes c(7) involved in Fe(III) respiration by Geobacter sulfurreducens.
Pokkuluri, P.R., Londer, Y.Y., Yang, X., Duke, N.E., Erickson, J., Orshonsky, V., Johnson, G., Schiffer, M.(2010) Biochim Biophys Acta 1797: 222-232
- PubMed: 19857457
- DOI: https://doi.org/10.1016/j.bbabio.2009.10.007
- Primary Citation of Related Structures:
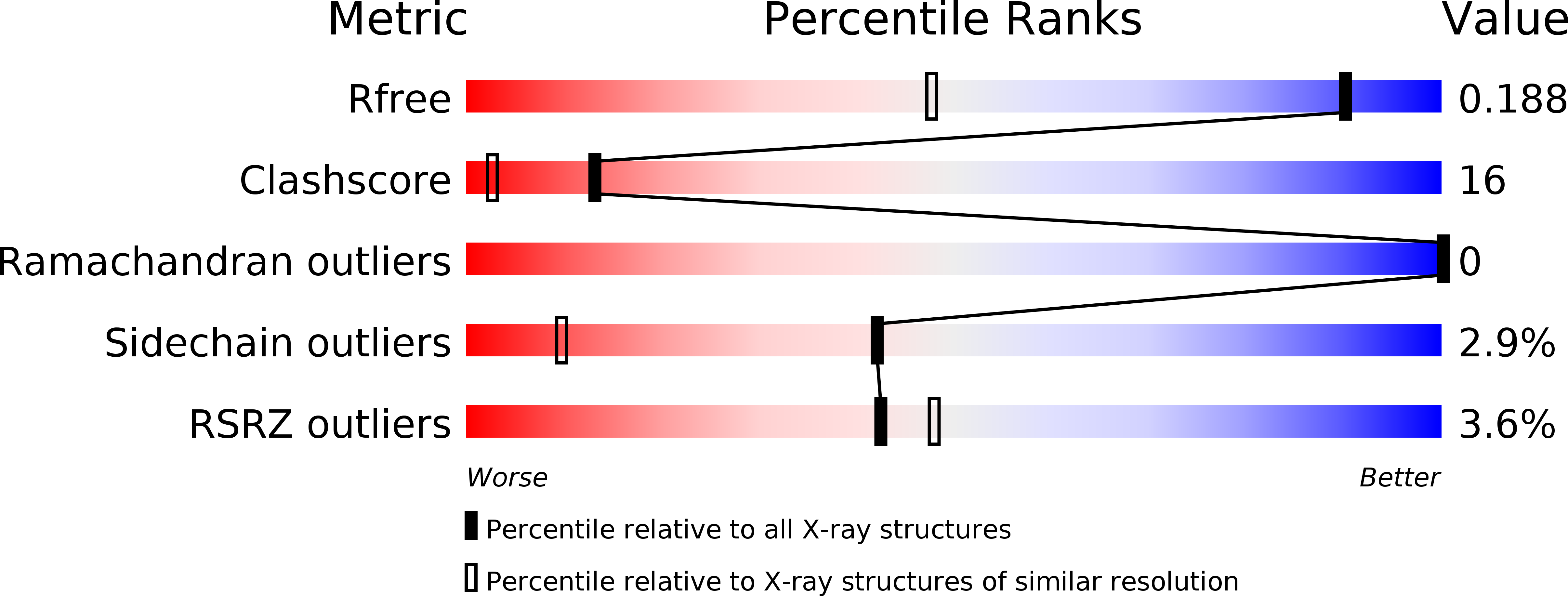
3H33, 3H34, 3H4N - PubMed Abstract:
Periplasmic cytochromes c(7) are important in electron transfer pathway(s) in Fe(III) respiration by Geobacter sulfurreducens. The genome of G. sulfurreducens encodes a family of five 10-kDa, three-heme cytochromes c(7). The sequence identity between the five proteins (designated PpcA, PpcB, PpcC, PpcD, and PpcE) varies between 45% and 77%. Here, we report the high-resolution structures of PpcC, PpcD, and PpcE determined by X-ray diffraction. This new information made it possible to compare the sequences and structures of the entire family. The triheme cores are largely conserved but are not identical. We observed changes, due to different crystal packing, in the relative positions of the hemes between two molecules in the crystal. The overall protein fold of the cytochromes is similar. The structure of PpcD differs most from that of the other homologs, which is not obvious from the sequence comparisons of the family. Interestingly, PpcD is the only cytochrome c(7) within the family that has higher abundance when G. sulfurreducens is grown on insoluble Fe(III) oxide compared to ferric citrate. The structures have the highest degree of conservation around "heme IV"; the protein surface around this heme is positively charged in all of the proteins, and therefore all cytochromes c(7) could interact with similar molecules involving this region. The structures and surface characteristics of the proteins near the other two hemes, "heme I" and "heme III", differ within the family. The above observations suggest that each of the five cytochromes c(7) could interact with its own redox partner via an interface involving the regions of heme I and/or heme III; this provides a possible rationalization for the existence of five similar proteins in G. sulfurreducens.
Organizational Affiliation:
Biosciences Division, Argonne National Laboratory, Argonne, IL 60439, USA.