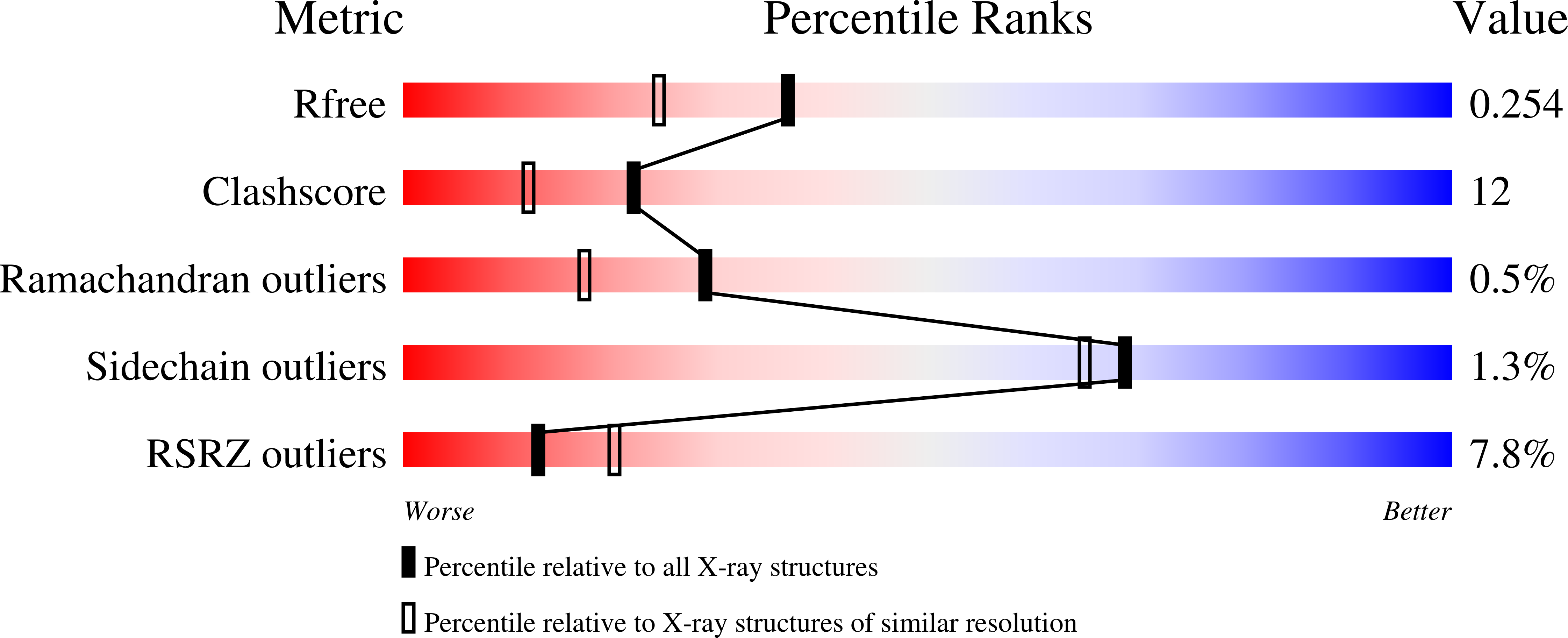
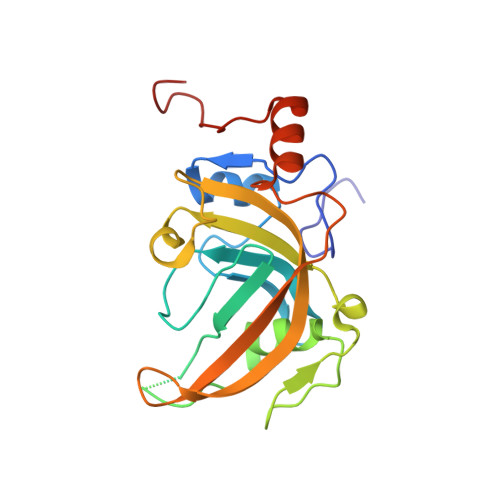
Structure and Hemimethylated CpG Binding of the SRA Domain from Human UHRF1.
Qian, C., Li, S., Jakoncic, J., Zeng, L., Walsh, M.J., Zhou, M.M.(2008) J Biol Chem 283: 34490-34494
- PubMed: 18945682
- DOI: https://doi.org/10.1074/jbc.C800169200
- Primary Citation of Related Structures:
3DWH - PubMed Abstract:
Human UHRF1 (ubiquitin-like PHD and RING finger 1) functions to maintain CpG DNA methylation patterns through DNA replication by co-localizing with the DNA methyltransferase DNMT1 at chromatin in mammals. Recent studies show that UHRF1 binds selectively to hemimethylated CpG via its conserved SRA (SET- and RING finger-associated) domain. However, the underlying molecular mechanism is not known. Here, we report a 1.95 A resolution crystal structure of the SRA domain of human UHRF1. Using NMR structure-guided mutagenesis, electrophoretic mobility shift assay, and fluorescence anisotropy analysis, we determined key amino acid residues for methyl-DNA binding that are conserved in the SRA domain.
Organizational Affiliation:
Department of Structural and Chemical Biology, Mount Sinai School of Medicine, New York University, New York, New York 10029, USA.