One residue substitution in PcyA leads to unexpected changes in tetrapyrrole substrate binding.
Wada, K., Hagiwara, Y., Yutani, Y., Fukuyama, K.(2010) Biochem Biophys Res Commun 402: 373-377
- PubMed: 20946883
- DOI: https://doi.org/10.1016/j.bbrc.2010.10.037
- Primary Citation of Related Structures:
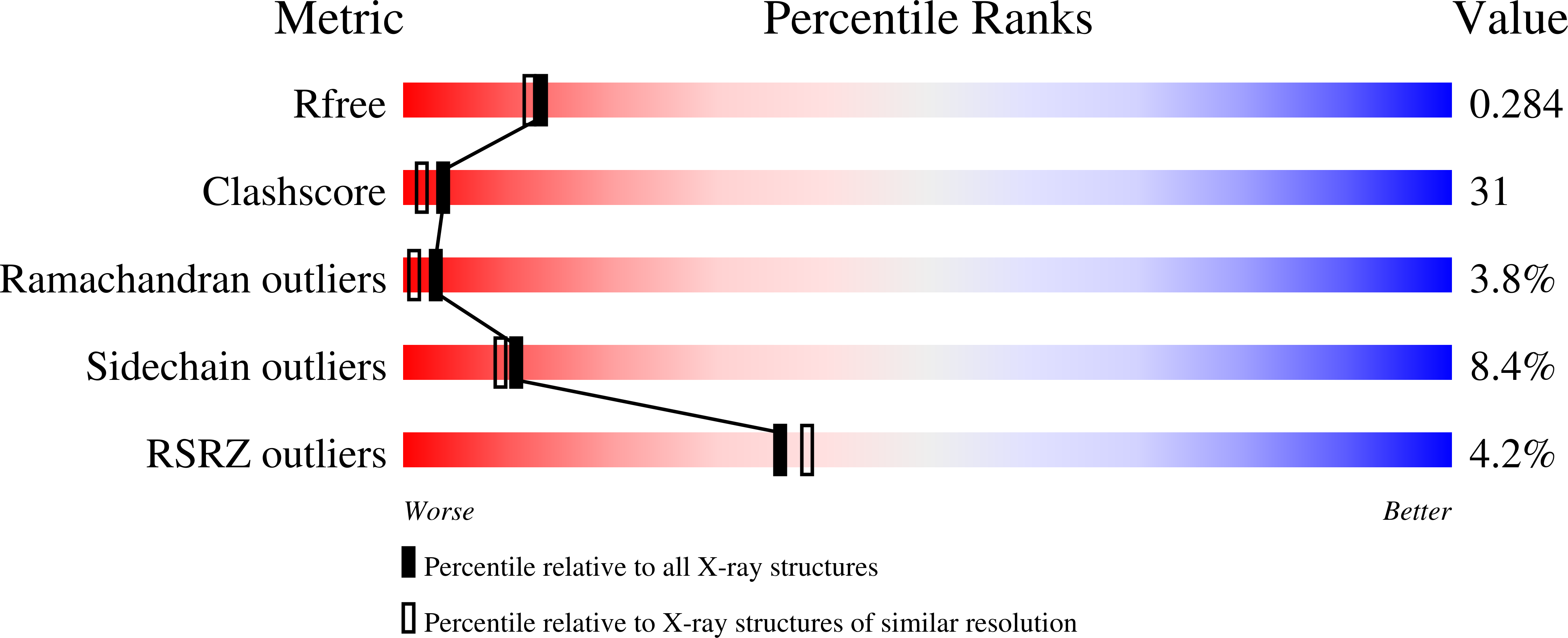
3AJG, 3AJH - PubMed Abstract:
Phycocyanobilin:ferredoxin oxidoreductase (PcyA) catalyzes the sequential reduction of the vinyl group of the D-ring and A-ring of biliverdin IXα (BV), using reducing equivalents provided by ferredoxin. This reaction produces phycocyanobilin, a pigment used for light-harvesting and light-sensing in red algae and cyanobacteria. The crystal structure of PcyA-BV reveals that BV is specifically bound in the PcyA active pocket through extensive hydrophobic and hydrophilic interactions. During the course of a mutational study of PcyA, we observed that mutation of the V225 position, apart from the processing sites, conferred an unusual property on PcyA; V225D mutant protein could bind BV and its analog BV13, but these complexes showed a distinct UV-vis absorption spectrum from that of the wild-type PcyA-BV complex. The crystal structures of BV- and BV13-bound forms of V225D protein revealed that gross structural changes occurred near the substrate-binding pocket, and that the BV/BV13 binding manner in the pocket was dramatically altered. Protein folding in V225D-BV/BV13 was more similar to that of substrate-free PcyA than that in PcyA-BV; the "induced-fit" did not occur when BV/BV13 was bound to the V225D protein. The unexpected structural change presented here provides a cautionary note about interpreting functional data derived from a mutated protein in the absence of its exact structure.
Organizational Affiliation:
Department of Biological Sciences, Graduate School of Science, Osaka University, Toyonaka, Osaka 560-0043, Japan.