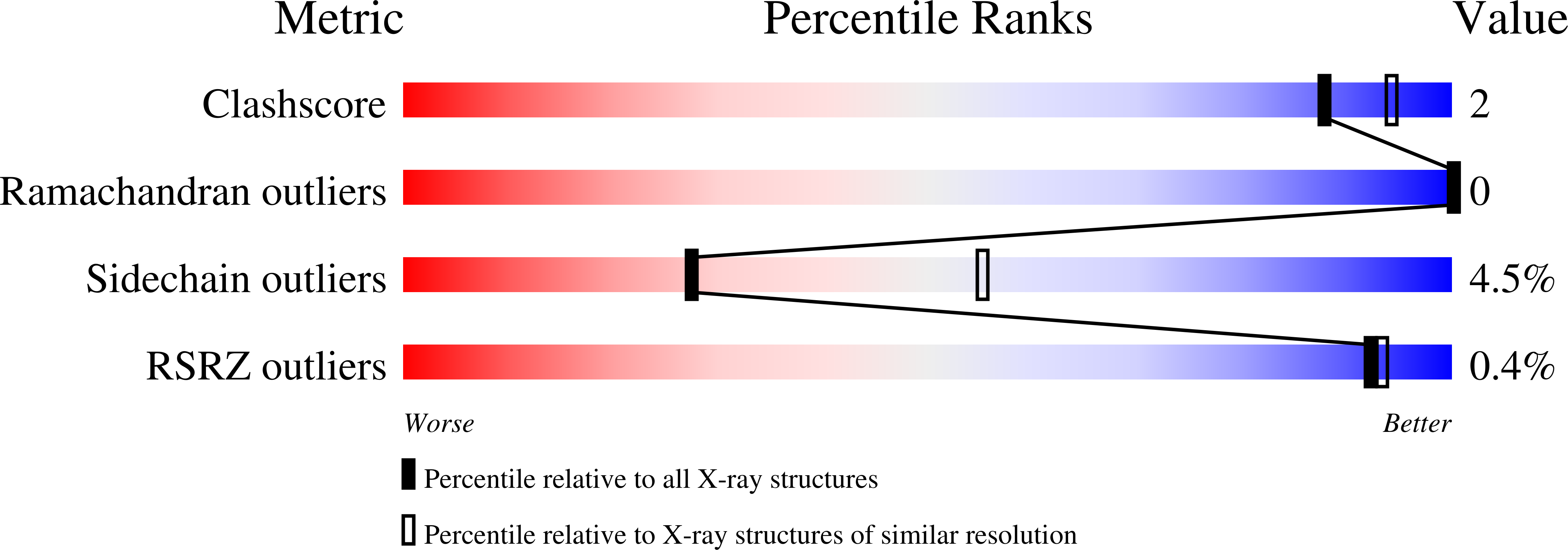
Screening a Limited Structure-Based Library Identifies Udp-Galnac-Specific Mutants of {Alpha}-1,3-Galactosyltransferase.
Tumbale, P., Jamaluddin, H., Thiyagarajan, N., Acharya, K.R., Brew, K.(2008) Glycobiology 18: 1036
- PubMed: 18782853
- DOI: https://doi.org/10.1093/glycob/cwn083
- Primary Citation of Related Structures:
2VXL, 2VXM - PubMed Abstract:
Complex glycans have important roles in biological recognition processes and considerable pharmaceutical potential. The synthesis of novel glycans can be facilitated by engineering glycosyltransferases to modify their substrate specificities. The choice of sites to modify requires the knowledge of the structures of enzyme-substrate complexes while the complexity of protein structures necessitates the exploration of a large array of multisite mutations. The retaining glycosyltransferase, alpha-1,3-galactosyltransferase (alpha3GT), which catalyzes the synthesis of the alpha-Gal epitope, has strict specificity for UDP-galactose as a donor substrate. Based on the structure of a complex of UDP-galactose with alpha3GT, the specificity for the galactose moiety can be partly attributed to residues that interact with the galactose 2-OH group, particularly His280 and Ala282. With the goal of engineering a variant of bovine alpha3GT with GalNAc transferase activity, we constructed a limited library of 456 alpha3GT mutants containing 19 alternative amino acids at position 280, two each at 281 and 282 and six at position 283. Clones (1500) were screened by assaying partially purified bacterially expressed variants for GalNAc transferase activity. Mutants with the highest levels of GalNAc transferase activity, AGGL or GGGL, had substitutions at all four sites. The AGGL mutant had slightly superior GalNAc transferase activity amounting to about 3% of the activity of the wild-type enzyme with UDP-Gal. This mutant had a low activity with UDP-Gal; its crystallographic structure suggests that the smaller side chains at residues 280-282 form a pocket to accommodate the larger acetamido group of GalNAc. Mutational studies indicate that Leu283 is important for stability in this mutant.
Organizational Affiliation:
Department of Biomedical Science, College of Biomedical Science, Florida Atlantic University, Glades Road, Boca Raton, FL 33431, USA.