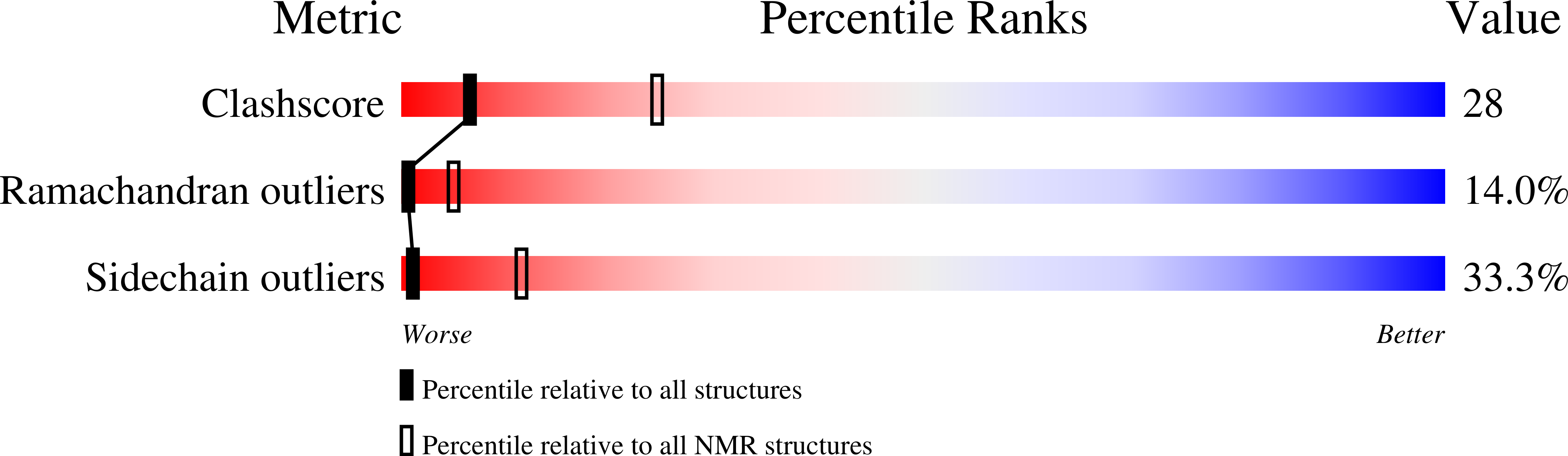Eukaryotic N-Glycosylation Occurs via the Membrane-anchored C-terminal Domain of the Stt3p Subunit of Oligosaccharyltransferase.
Huang, C., Bhaskaran, R., Mohanty, S.(2012) J Biol Chem 287: 32450-32458
- PubMed: 22865878
- DOI: https://doi.org/10.1074/jbc.M112.342253
- Primary Citation of Related Structures:
2LGZ - PubMed Abstract:
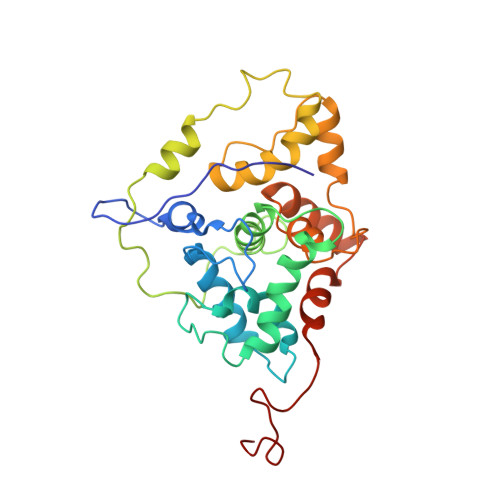
N-glycosylation is an essential and highly conserved protein modification. In eukaryotes, it is catalyzed by a multisubunit membrane-associated enzyme, oligosaccharyltransferase (OT). We report the high resolution structure of the C-terminal domain of eukaryotic Stt3p. Unlike its soluble β-sheet-rich prokaryotic counterparts, our model reveals that the C-terminal domain of yeast Stt3p is highly helical and has an overall oblate spheroid-shaped structure containing a membrane-embedded region. Anchoring of this protein segment to the endoplasmic reticulum membrane is likely to bring the membrane-embedded donor substrate closer, thus facilitating glycosylation efficiency. Structural comparison of the region near the WWDYG signature motif revealed that the acceptor substrate-binding site of yeast OT strikingly resembles its prokaryotic counterparts, suggesting a conserved mechanism of N-glycosylation from prokaryotes to eukaryotes. Furthermore, comparison of the NMR and cryo-EM structures of yeast OT revealed that the molecular architecture of this acceptor substrate-recognizing domain has interesting spatial specificity for interactions with other essential OT subunits.
Organizational Affiliation:
Department of Chemistry and Biochemistry, Auburn University, Auburn, Alabama 36849, USA.