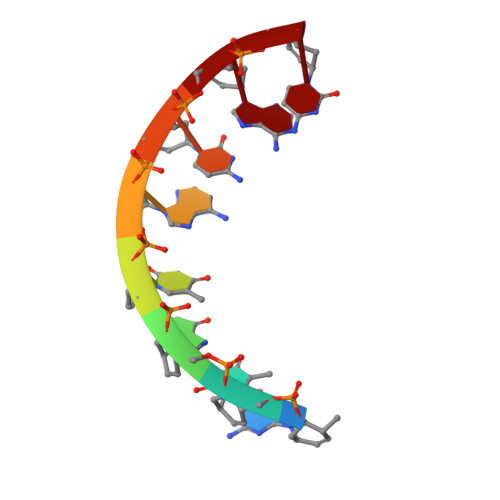
Structure of the fully modified left-handed cyclohexene nucleic acid sequence GTGTACAC.
Robeyns, K., Herdewijn, P., Van Meervelt, L.(2008) J Am Chem Soc 130: 1979-1984
- PubMed: 18198873
- DOI: https://doi.org/10.1021/ja077313f
- Primary Citation of Related Structures:
2H0N - PubMed Abstract:
CeNA oligonucleotides consist of a phosphorylated backbone where the deoxyribose sugars are replaced by cyclohexene moieties. The X-ray structure determination and analysis of a fully modified octamer sequence GTGTACAC, which is the first crystal structure of a carbocyclic-based nucleic acid, is presented. This particular sequence was built with left-handed building blocks and crystallizes as a left-handed double helix. The helix can be characterized as belonging to the (mirrored) A-type family. Crystallographic data were processed up to 1.53 A, and the octamer sequence crystallizes in the space group R32. The sugar puckering is found to adopt the 3H2 half-chair conformation which mimics the C3'-endo conformation of the ribose sugar. The double helices stack on top of each other to form continuous helices, and static disorder is observed due to this end-to-end stacking.
Organizational Affiliation:
Katholieke Universiteit Leuven, Department of Chemistry, Biomolecular Architecture and BioMacS, Celestijnenlaan 200F, B-3001 Leuven, Belgium.