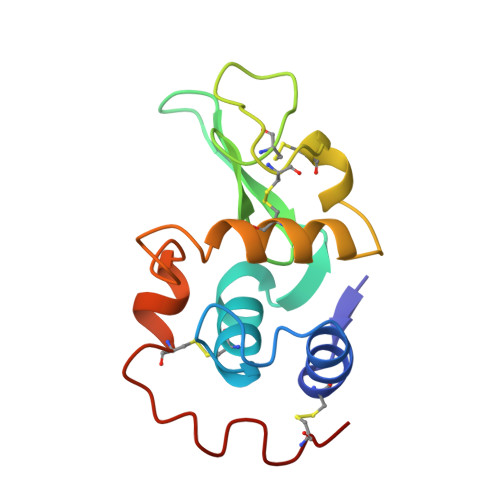
Crystallographic studies of a calcium binding lysozyme from equine milk at 2.5 A resolution.
Tsuge, H., Ago, H., Noma, M., Nitta, K., Sugai, S., Miyano, M.(1992) J Biochem 111: 141-143
- PubMed: 1569037
- DOI: https://doi.org/10.1093/oxfordjournals.jbchem.a123727
- Primary Citation of Related Structures:
2EQL - PubMed Abstract:
The crystal structure of a calcium binding equine lysozyme has been determined at 2.5 A resolution by means of molecular replacement. The energy minimized equine lysozyme as the starting model, was refined with the molecular dynamics program, X-PLOR, and the R factor of the current model was found to be 24% without any water molecules. The conformation of the calcium binding loop is similar to that of alpha-lactalbumin. The profiles of backbone atomic displacements throughout the lysozyme and alpha-lactalbumin superfamilies are comparable as well as their homologous tertiary structures.
Organizational Affiliation:
Life Science Research Laboratory, Japan Tobacco, Inc., Kanagawa.