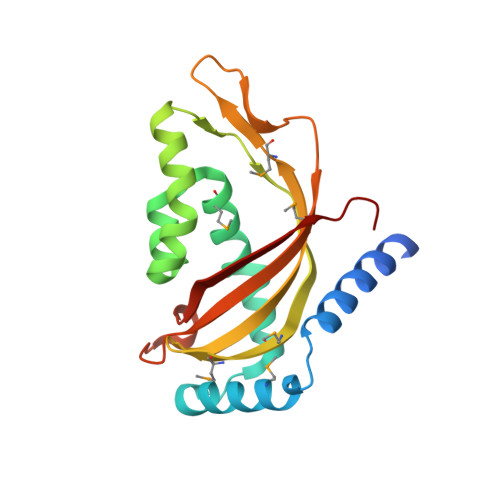
Structure of the human Tim44 C-terminal domain in complex with pentaethylene glycol: ligand-bound form.
Handa, N., Kishishita, S., Morita, S., Akasaka, R., Jin, Z., Chrzas, J., Chen, L., Liu, Z.J., Wang, B.C., Sugano, S., Tanaka, A., Terada, T., Shirouzu, M., Yokoyama, S.(2007) Acta Crystallogr D Biol Crystallogr 63: 1225-1234
- PubMed: 18084070
- DOI: https://doi.org/10.1107/S0907444907051463
- Primary Citation of Related Structures:
2CW9 - PubMed Abstract:
Familial oncocytic thyroid carcinoma is associated with a missense mutation, P308Q, in the C-terminal domain of Tim44. Tim44 is the mitochondrial inner-membrane translocase subunit and it functions as a membrane anchor for the mitochondrial heat-shock protein 70 (mtHsp70). Here, the crystal structure of the human Tim44 C-terminal domain complexed with pentaethylene glycol has been determined at 1.9 A resolution. The overall structure resembles that of the nuclear transport factor 2-like domain. In the crystal structure, pentaethylene glycol molecules are associated at two potential membrane-binding sites: the large hydrophobic cavity and the highly conserved loop between the alpha1 and alpha2 helices near Pro308. A comparison with the yeast homolog revealed that lipid binding induces conformational changes around the alpha1-alpha2 loop, leading to slippage of the alpha1 helix along the large beta-sheet. These changes may play important roles in the translocation of polypeptides across the mitochondrial inner membrane.
Organizational Affiliation:
Protein Research Group, Genomic Sciences Center, Yokohama Institute, RIKEN, 1-7-22 Suehiro, Tsurumi, Yokohama 230-0045, Japan.