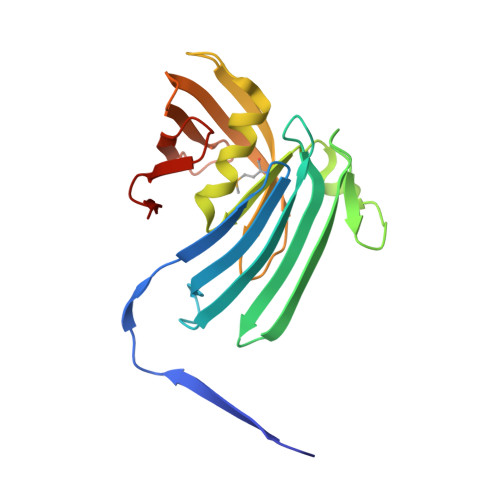
The structure of the first representative of Pfam family PF06475 reveals a new fold with possible involvement in glycolipid metabolism.
Bakolitsa, C., Kumar, A., McMullan, D., Krishna, S.S., Miller, M.D., Carlton, D., Najmanovich, R., Abdubek, P., Astakhova, T., Chiu, H.J., Clayton, T., Deller, M.C., Duan, L., Elias, Y., Feuerhelm, J., Grant, J.C., Grzechnik, S.K., Han, G.W., Jaroszewski, L., Jin, K.K., Klock, H.E., Knuth, M.W., Kozbial, P., Marciano, D., Morse, A.T., Nigoghossian, E., Okach, L., Oommachen, S., Paulsen, J., Reyes, R., Rife, C.L., Trout, C.V., van den Bedem, H., Weekes, D., White, A., Xu, Q., Hodgson, K.O., Wooley, J., Elsliger, M.A., Deacon, A.M., Godzik, A., Lesley, S.A., Wilson, I.A.(2010) Acta Crystallogr Sect F Struct Biol Cryst Commun 66: 1211-1217
- PubMed: 20944213
- DOI: https://doi.org/10.1107/S1744309109022684
- Primary Citation of Related Structures:
2H1T - PubMed Abstract:
The crystal structure of PA1994 from Pseudomonas aeruginosa, a member of the Pfam PF06475 family classified as a domain of unknown function (DUF1089), reveals a novel fold comprising a 15-stranded β-sheet wrapped around a single α-helix that assembles into a tight dimeric arrangement. The remote structural similarity to lipoprotein localization factors, in addition to the presence of an acidic pocket that is conserved in DUF1089 homologs, phospholipid-binding and sugar-binding proteins, indicate a role for PA1994 and the DUF1089 family in glycolipid metabolism. Genome-context analysis lends further support to the involvement of this family of proteins in glycolipid metabolism and indicates possible activation of DUF1089 homologs under conditions of bacterial cell-wall stress or host-pathogen interactions.
Organizational Affiliation:
Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, Menlo Park, CA, USA.