The 2 a Crystal Structure of 6-Oxo Camphor Hydrolase: New Structural Diversity in the Crotonase Superfamily
Whittingham, J.L., Turkenburg, J.P., Verma, C.S., Walsh, M.A., Grogan, G.(2003) J Biol Chem 278: 1744
- PubMed: 12421807
- DOI: https://doi.org/10.1074/jbc.M211188200
- Primary Citation of Related Structures:
1O8U - PubMed Abstract:
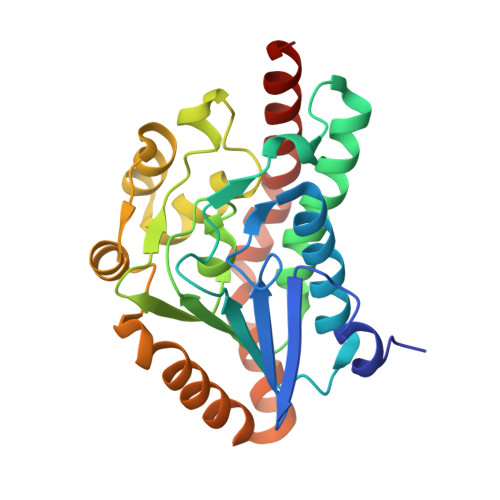
6-Oxo camphor hydrolase (OCH) is an enzyme of the crotonase superfamily that catalyzes carbon-carbon bond cleavage in bicyclic beta-diketones via a retro-Claisen reaction (Grogan, G., Roberts, G. A., Bougioukou, D., Turner, N. J., and Flitsch, S. L. (2001) J. Biol. Chem. 276, 12565-12572). The native structure of OCH has been solved at 2.0-A resolution with selenomethionine multiple wave anomalous dispersion and refined to a final R(free) of 19.0. The structure of OCH consists of a dimer of trimers that resembles the "parent" enzyme of the superfamily, enoyl-CoA hydratase. In contrast to enoyl-CoA hydratase, however, two octahedrally coordinated sodium atoms are found at the 3-fold axis of the hexamer of OCH, and the C-terminal helix of OCH does not form a discrete domain. Models of the substrate, 6-oxo camphor, and a proposed enolate intermediate in the putative active site suggest possible mechanistic roles for Glu-244, Asp-154, His-122, His-45, and His-145.
Organizational Affiliation:
York Structural Biology Laboratory, Department of Chemistry, University of York, Heslington, York, North Yorkshire YO10 5YW, United Kingdom.