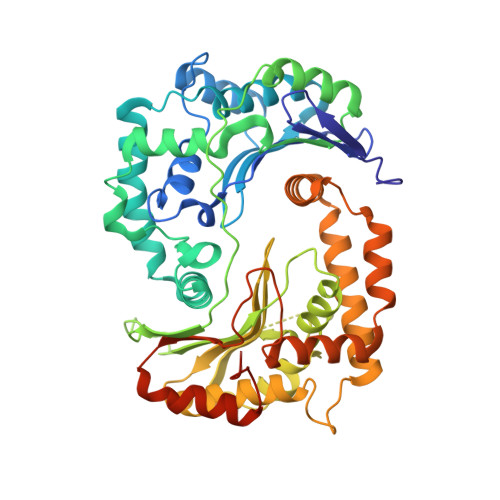
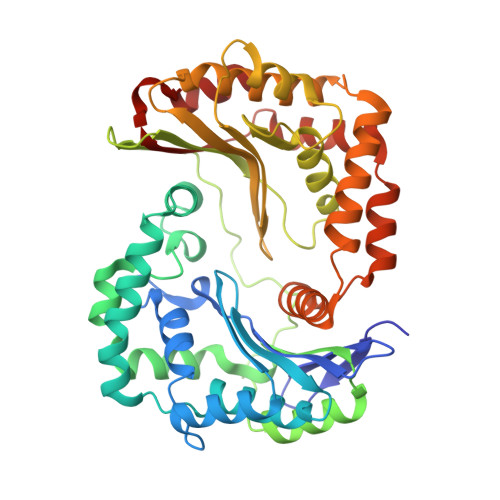
Crystal structures of mitochondrial processing peptidase reveal the mode for specific cleavage of import signal sequences.
Taylor, A.B., Smith, B.S., Kitada, S., Kojima, K., Miyaura, H., Otwinowski, Z., Ito, A., Deisenhofer, J.(2001) Structure 9: 615-625
- PubMed: 11470436
- DOI: https://doi.org/10.1016/s0969-2126(01)00621-9
- Primary Citation of Related Structures:
1HR6, 1HR7, 1HR8, 1HR9 - PubMed Abstract:
Mitochondrial processing peptidase (MPP) is a metalloendopeptidase that cleaves the N-terminal signal sequences of nuclear-encoded proteins targeted for transport from the cytosol to the mitochondria. Mitochondrial signal sequences vary in length and sequence, but each is cleaved at a single specific site by MPP. The cleavage sites typically contain an arginine at position -2 (in the N-terminal portion) from the scissile peptide bond in addition to other distal basic residues, and an aromatic residue at position +1. Mitochondrial import machinery recognizes amphiphilic helical conformations in signal sequences. However, it is unclear how MPP specifically recognizes diverse presequence substrates.
Organizational Affiliation:
Howard Hughes Medical Institute, University of Texas Southwestern Medical Center, Dallas, TX 75390, USA.