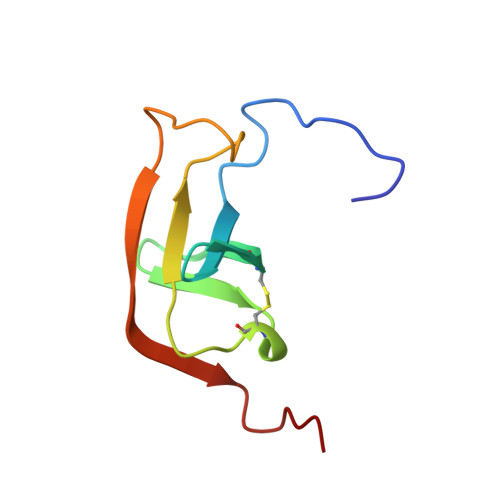
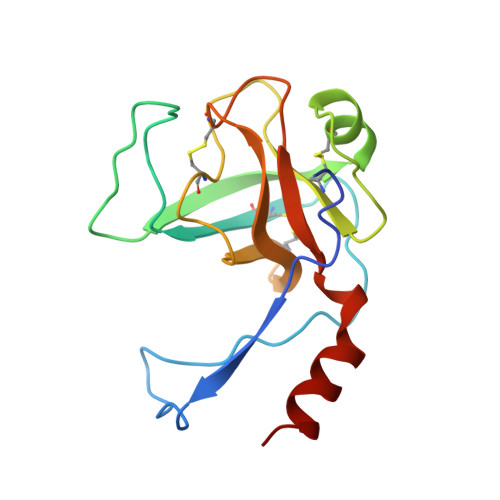
A new structural class of serine protease inhibitors revealed by the structure of the hirustasin-kallikrein complex.
Mittl, P.R., Di Marco, S., Fendrich, G., Pohlig, G., Heim, J., Sommerhoff, C., Fritz, H., Priestle, J.P., Grutter, M.G.(1997) Structure 5: 253-264
- PubMed: 9032072
- DOI: https://doi.org/10.1016/s0969-2126(97)00183-4
- Primary Citation of Related Structures:
1HIA - PubMed Abstract:
Hirustasin belongs to a class of serine protease inhibitors characterized by a well conserved pattern of cysteine residues. Unlike the closely related inhibitors, antistasin/ghilanten and guamerin, which are selective for coagulation factor Xa or neutrophil elastase, hirustasin binds specifically to tissue kallikrein. The conservation of the pattern of cysteine residues and the significant sequence homology suggest that these related inhibitors possess a similar three-dimensional structure to hirustasin.
Organizational Affiliation:
CDDT Pharmaceutical Research, Ciba-Geigy Ltd., 4002 Basel, Switzerland.