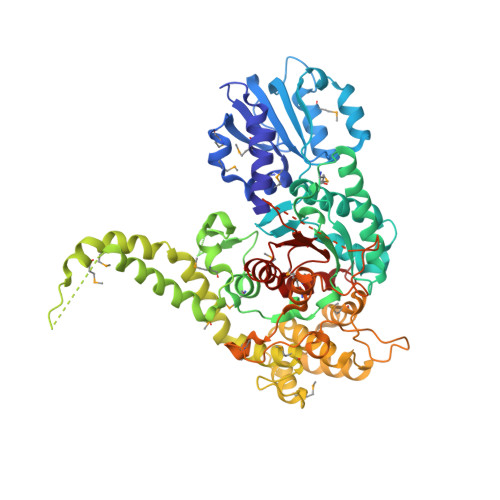
The X-ray crystal structure of neuronal Sec1 from squid sheds new light on the role of this protein in exocytosis.
Bracher, A., Perrakis, A., Dresbach, T., Betz, H., Weissenhorn, W.(2000) Structure 8: 685-694
- PubMed: 10903948
- DOI: https://doi.org/10.1016/s0969-2126(00)00156-8
- Primary Citation of Related Structures:
1EPU - PubMed Abstract:
Sec1-like molecules have been implicated in a variety of eukaryotic vesicle transport processes including neurotransmitter release by exocytosis. They regulate vesicle transport by binding to a t-SNARE from the syntaxin family. This process is thought to prevent SNARE complex formation, a protein complex required for membrane fusion. Whereas Sec1 molecules are essential for neurotransmitter release and other secretory events, their interaction with syntaxin molecules seems to represent a negative regulatory step in secretion.
Organizational Affiliation:
European Molecular Biology Laboratory (EMBL), Grenoble, 38000, France.