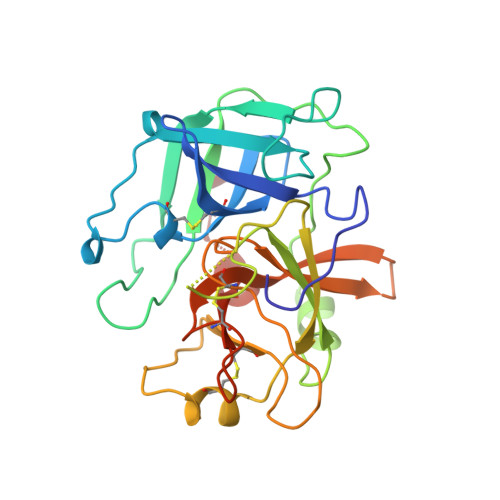
Enzyme flexibility, solvent and 'weak' interactions characterize thrombin-ligand interactions: implications for drug design.
Engh, R.A., Brandstetter, H., Sucher, G., Eichinger, A., Baumann, U., Bode, W., Huber, R., Poll, T., Rudolph, R., von der Saal, W.(1996) Structure 4: 1353-1362
- PubMed: 8939759
- DOI: https://doi.org/10.1016/s0969-2126(96)00142-6
- Primary Citation of Related Structures:
1UVS, 1UVT, 1UVU - PubMed Abstract:
The explosive growth in the rate of X-ray determination of protein structures is fuelled largely by the expectation that structural information will be useful for pharmacological and biotechnological applications. For example, there have been intensive efforts to develop orally administrable antithrombotic drugs using information about the crystal structures of blood coagulation factors, including thrombin. Most of the low molecular weight thrombin inhibitors studied so far are based on arginine and benzamidine. We sought to expand the database of information on thrombin-inhibitor binding by studying new classes of inhibitors. We report the structures of three new inhibitors complexed with thrombin, two based on 4-aminopyridine and one based on naphthamidine. We observe several geometry changes in the protein main chain and side chains which accompany inhibitor binding. The two inhibitors based on 4-aminopyridine bind in notably different ways: one forms a water-mediated hydrogen bond to the active site Ser195, the other induces a rotation of the Ser214-Trp215 peptide plane that is unprecedented in thrombin structures. These binding modes also differ in their 'weak' interactions, including CH-O hydrogen bonds and interactions between water molecules and aromatic pi-clouds. Induced-fit structural changes were also seen in the structure of the naphthamidine inhibitor complex. Protein flexibility and variable water structures are essential elements in protein-ligand interactions. Ligand design strategies that fail to take this into account may overlook or underestimate the potential of lead structures. Further, the significance of 'weak' interactions must be considered both in crystallographic refinement and in analysis of binding mechanisms.
Organizational Affiliation:
Max-Planck-Institut für Biochemie, D82152 Martinsried, Germany. engh@biochem.mpg.de