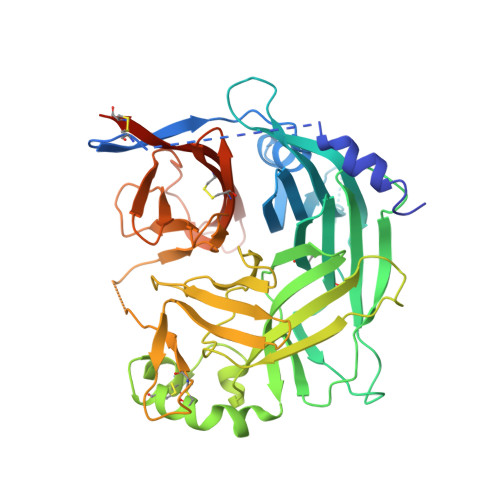
Crystal structure of measles virus hemagglutinin provides insight into effective vaccines
Hashiguchi, T., Kajikawa, M., Maita, N., Takeda, M., Kuroki, K., Sasaki, K., Kohda, D., Yanagi, Y., Maenaka, K.(2007) Proc Natl Acad Sci U S A 104: 19535-19540
- PubMed: 18003910
- DOI: https://doi.org/10.1073/pnas.0707830104
- Primary Citation of Related Structures:
2ZB5, 2ZB6 - PubMed Abstract:
Measles still remains a major cause of childhood morbidity and mortality worldwide. Measles virus (MV) vaccines are highly successful, but the mechanism underlying their efficacy has been unclear. Here we report the crystal structure of the MV attachment protein, hemagglutinin, responsible for MV entry. The receptor-binding head domain exhibits a cubic-shaped beta-propeller structure and forms a homodimer. N-linked sugars appear to mask the broad regions and cause the two molecules forming the dimer to tilt oppositely toward the horizontal plane. Accordingly, residues of the putative receptor-binding site, highly conserved among MV strains, are strategically positioned in the unshielded area of the protein. These conserved residues also serve as epitopes for neutralizing antibodies, ensuring the serological monotype, a basis for effective MV vaccines. Our findings suggest that sugar moieties in the MV hemagglutinin critically modulate virus-receptor interaction as well as antiviral antibody responses, differently from sugars of the HIV gp120, which allow for immune evasion.
Organizational Affiliation:
Department of Virology, Faculty of Medicine, and Division of Structural Biology, Medical Institute of Bioregulation, Kyushu University, 3-1-1 Maidashi, Higashi-ku, Fukuoka, Fukuoka 812-8582, Japan.