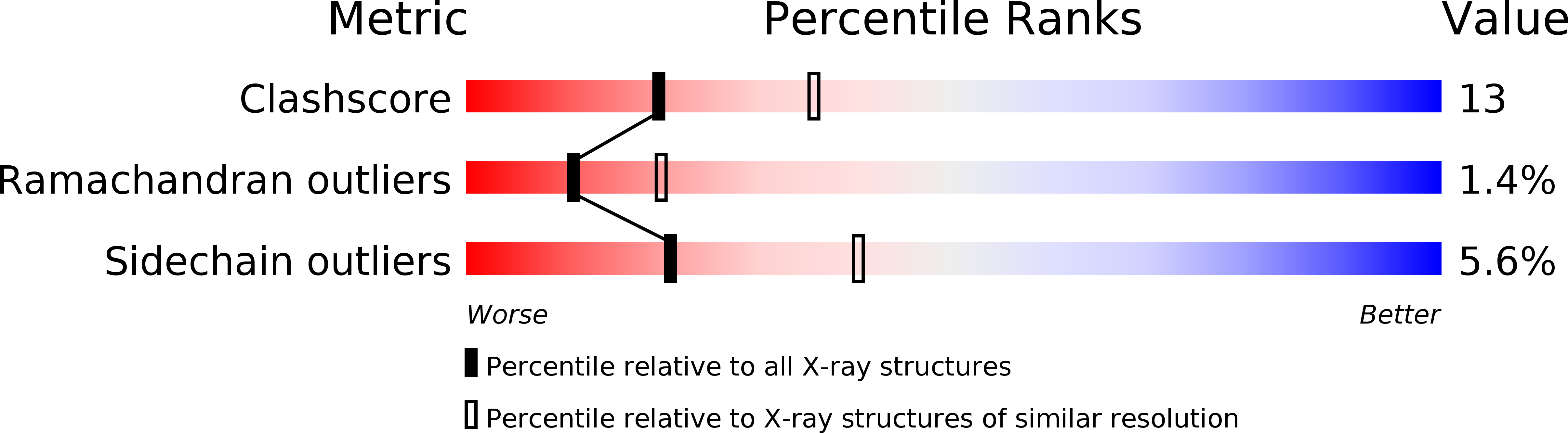Design, synthesis and biological evaluation of thrombin inhibitors based on a pyridine scaffold.
Blomberg, D., Fex, T., Xue, Y., Brickmann, K., Kihlberg, J.(2007) Org Biomol Chem 5: 2599-2605
- PubMed: 18019535
- DOI: https://doi.org/10.1039/b705344d
- Primary Citation of Related Structures:
2PKS - PubMed Abstract:
A series of 2,4-disubstituted pyridine derivatives has been designed, synthesised and evaluated as thrombin inhibitors. A Grignard exchange reaction was used to introduce various benzoyl substituents in position 4 of the pyridine ring, where they serve as P3 residues in binding to thrombin. In position 2 of the pyridine ring, a para-amidinobenzylamine moiety was incorporated as P1 residue by an SNAr reaction using ammonia as nucleophile followed by a reductive amination. A crystal structure obtained for one of the compounds in the active site of thrombin revealed that the basic amidine group of the inhibitor was anchored to Asp 189 at the bottom of the S1 pocket. A comparison with melagatran, bound in the active site of thrombin, revealed a good shape match but lack of hydrogen bonding possibilities in the S2-S3 region for the thrombin inhibitors reported in this study.
Organizational Affiliation:
Organic Chemistry, Department of Chemistry, Umeå University, SE-901 87 Umeå, Sweden.